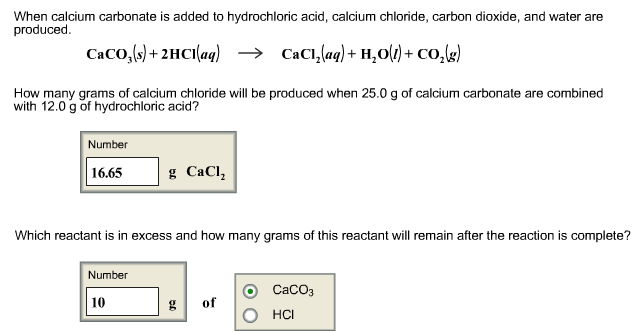
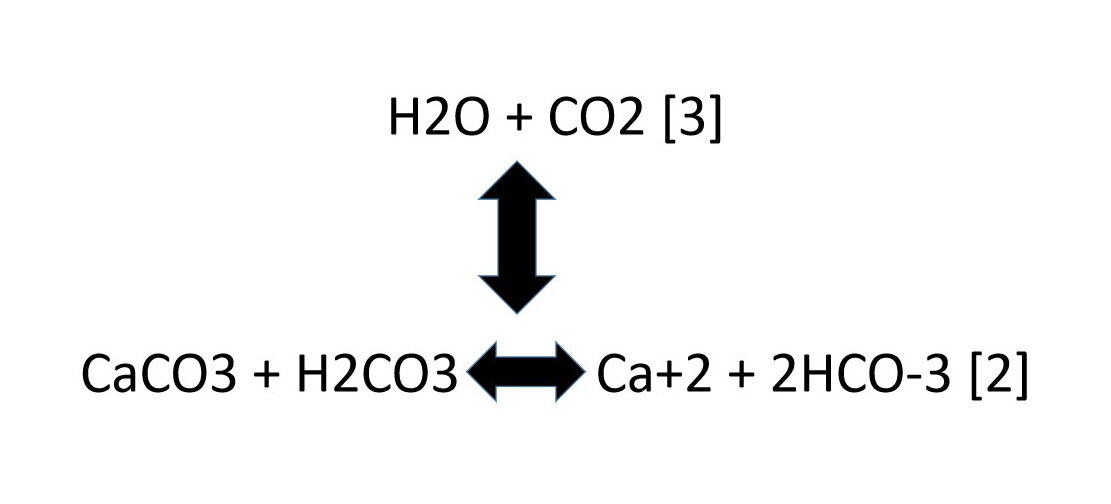
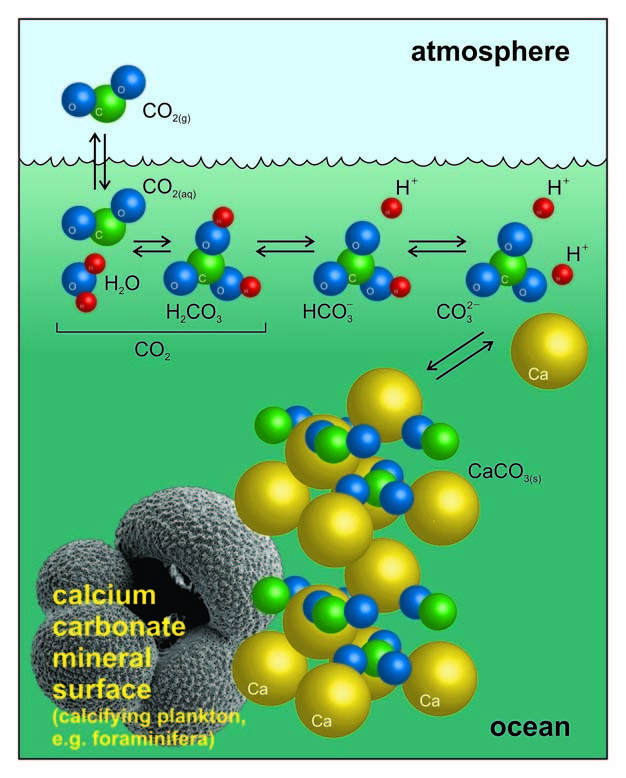
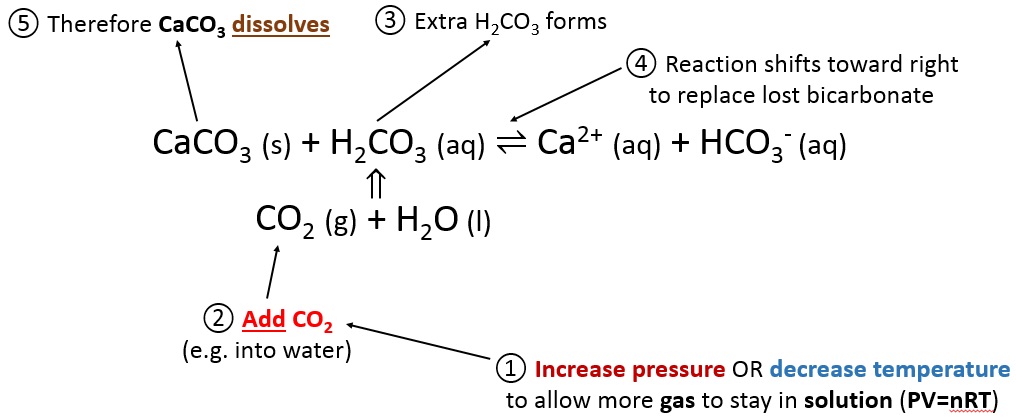
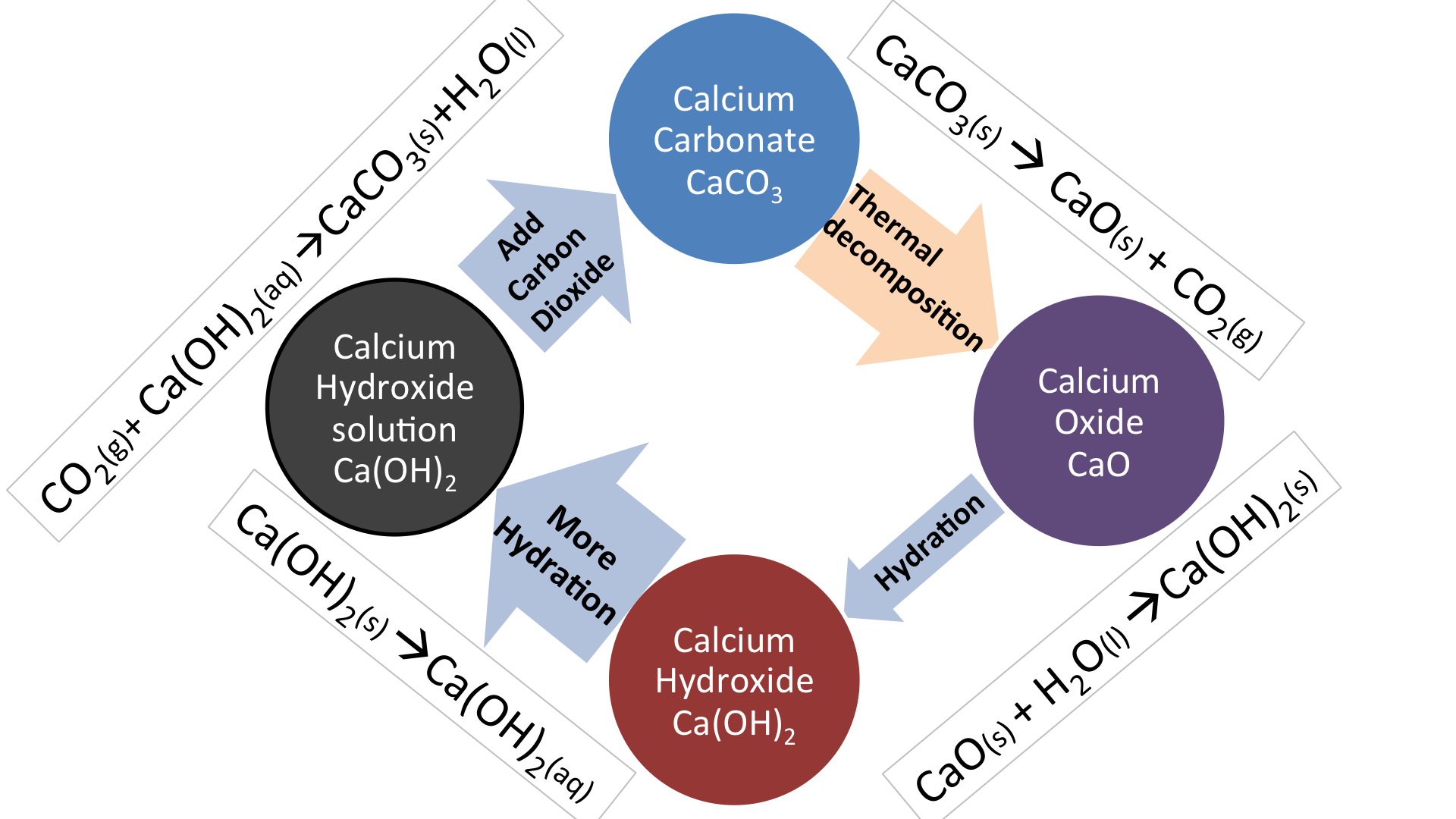
One method of determining the proportion of calcium carbonate in a coral is to dissolve a known mass of the coral in excess acid and measure the volume of carbon dioxide formed.
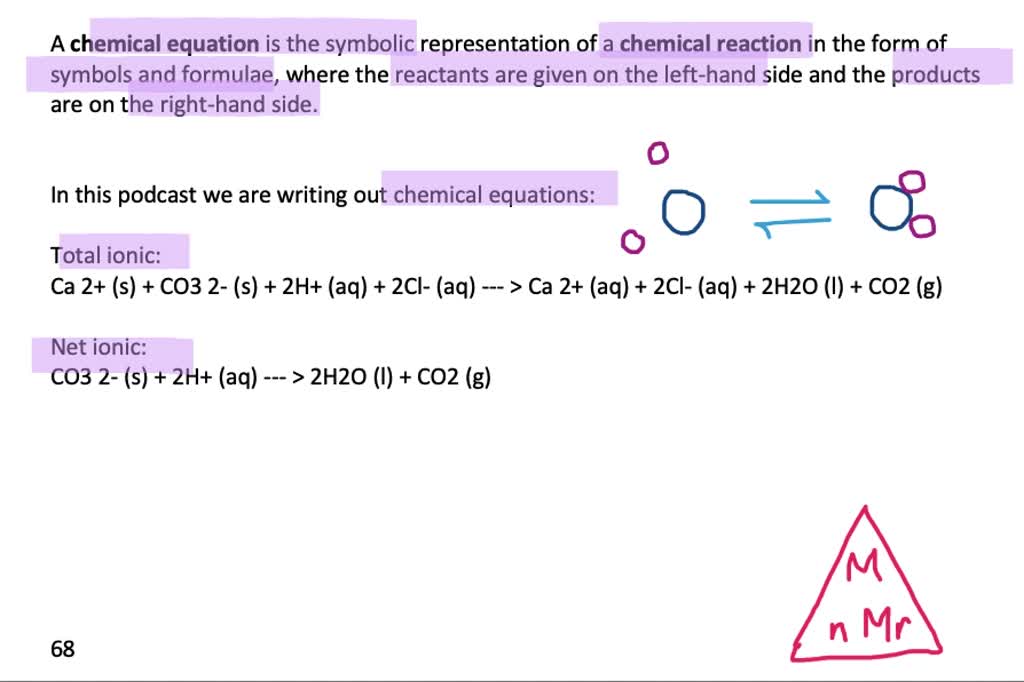
SOLVED:Limestone (calcium carbonate) is insoluble in water but dissolves when a hydrochloric acid solution is added. Write balanced total ionic and net ionic equations, showing hydrochloric acid as it actually exists in