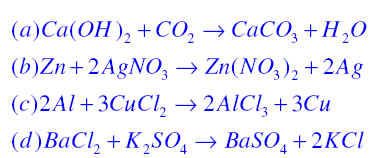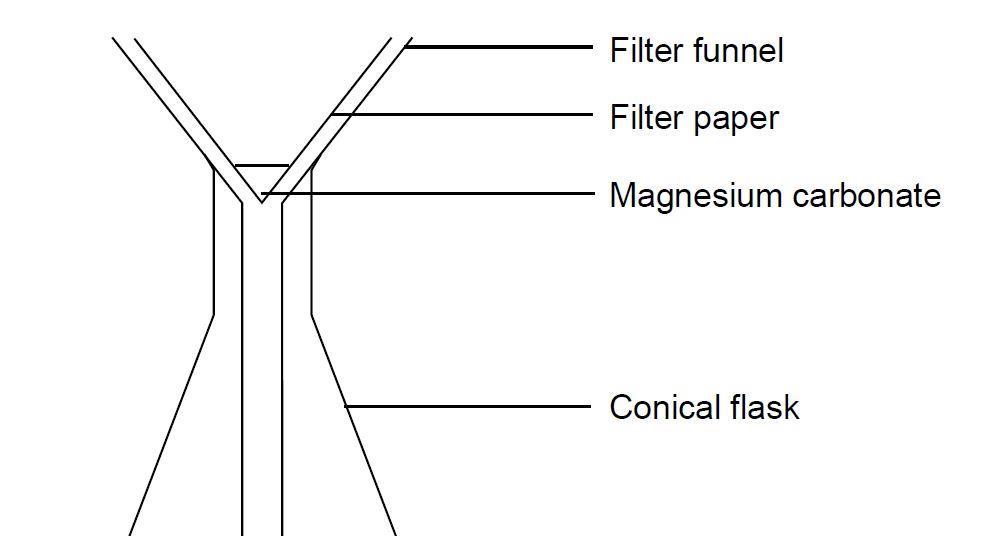
Making magnesium carbonate: the formation of an insoluble salt in water | Experiment | RSC Education

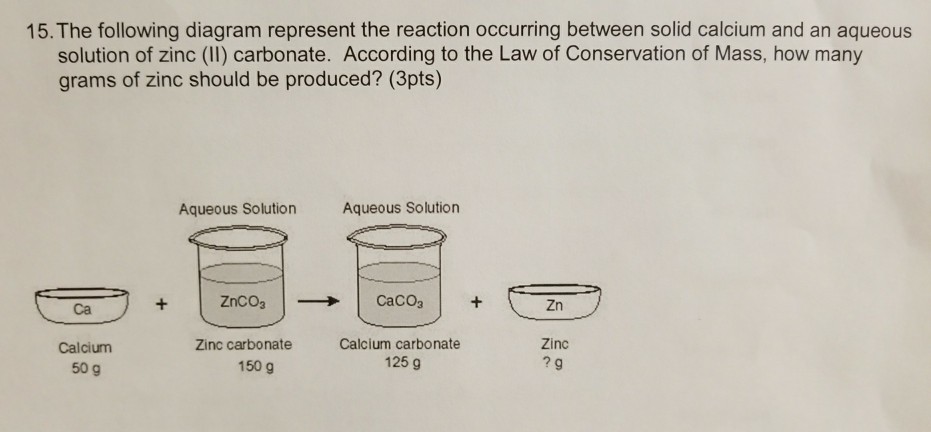
According to the law of conservation of mass, how much zinc was PRODUCED by reacting Calcium and Zinc - Brainly.com
Write the balanced chemical equations for the following reactions: i. Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water - Sarthaks eConnect | Largest Online Education Community

Write the balanced chemical equations for the following reactions.(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water(b) Zinc + Silver nitrate → Zinc nitrate + Silver(c) Aluminium + Copper chloride

Decomposition Reaction Infographic Diagram With Example Of Zinc Carbonate Breaking Down By Heat To Zinc Oxide And Carbon Dioxide For Chemistry Science Education Poster Vector Royalty Free SVG, Cliparts, Vectors, And Stock
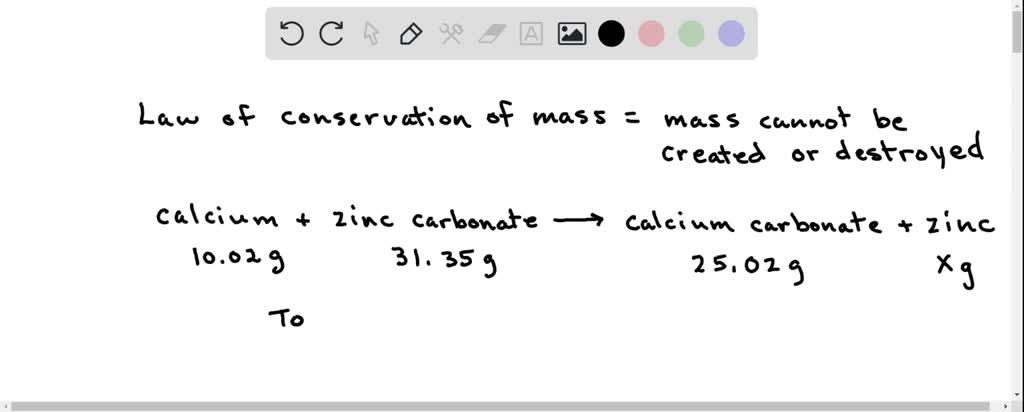
SOLVED: Calcium reacts with zinc carbonate to produce Calcium carbonate and Zinc. 31.35g of Zinc carbonate and 10.02g of calcium were reacted. The reaction produced 25.02g of calcium carbonate. What is the

PDF) The Effect of Zinc Oxide and Calcium Carbonate Nanoparticles on the Thermal Conductivity of Polypropylene

Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride
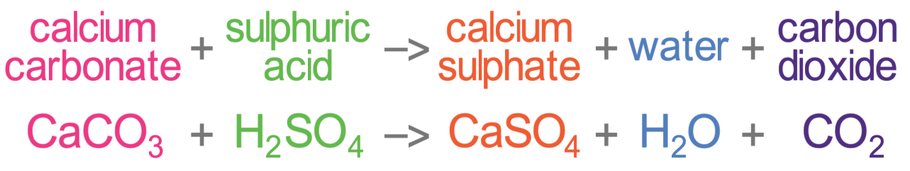
Making use only of substances given: dil. sulphuric acid, Sodium carbonate, Zinc, Sodium - Sarthaks eConnect | Largest Online Education Community

Mechanistic study of the synergetic inhibiting effects of Zn2+, Cu2+ and Mg2+ ions on calcium carbonate precipitation - ScienceDirect

Calcium carbonate reacts with zinc metal to produce 10g of zinc carbonate and calcium metal. Calculate the mass of zinc metal that reacted. | Homework.Study.com