![The hardness of a water sample (in terms of equivalents of CaCO3) containing 10^-3 M CaSO4 is:[Molar mass of CaSO4 = 136 g mol^-1] . The hardness of a water sample (in terms of equivalents of CaCO3) containing 10^-3 M CaSO4 is:[Molar mass of CaSO4 = 136 g mol^-1] .](https://dwes9vv9u0550.cloudfront.net/images/8020054/a6e110e8-cb36-4333-8440-cae1f5196351.jpg)
The hardness of a water sample (in terms of equivalents of CaCO3) containing 10^-3 M CaSO4 is:[Molar mass of CaSO4 = 136 g mol^-1] .

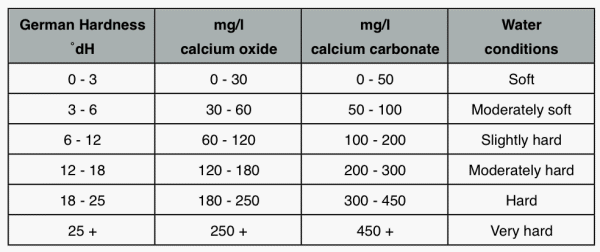
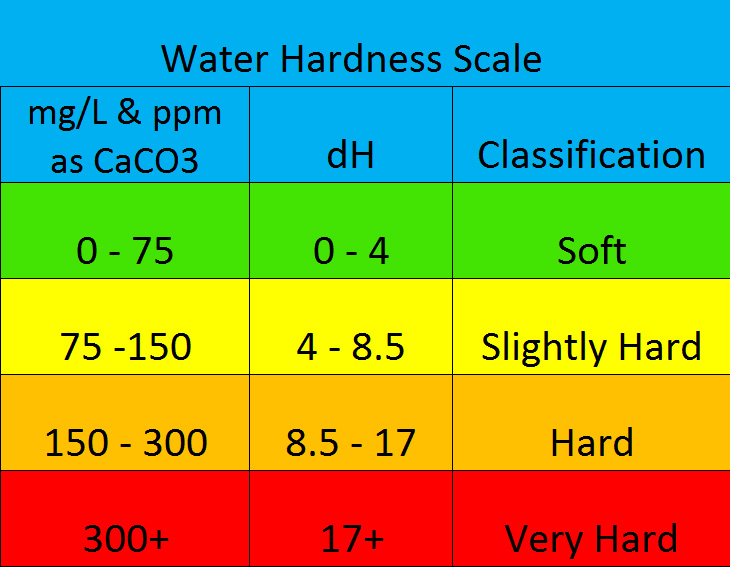
SOLVED: Although hardness can be caused by both calcium and magnesium ions, it is often expressed in terms of ppm CaCOz (mg CaCOz/L water) According to this scale, a hardness of 150

Shore A hardness of the NR composites with 40 phr of calcium carbonate... | Download Scientific Diagram

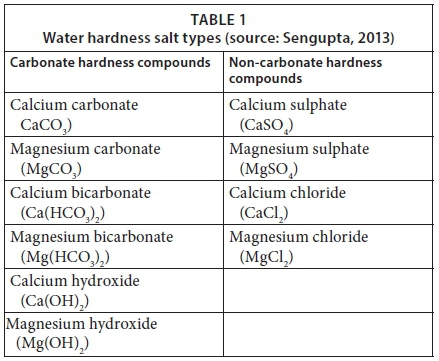
SOLVED: Calculating the Water Hardness For convenience assume that all hardness is due to the presence of only Caz+ions in the form of calcium carbonate, CaCO3 The unit of hardness may be

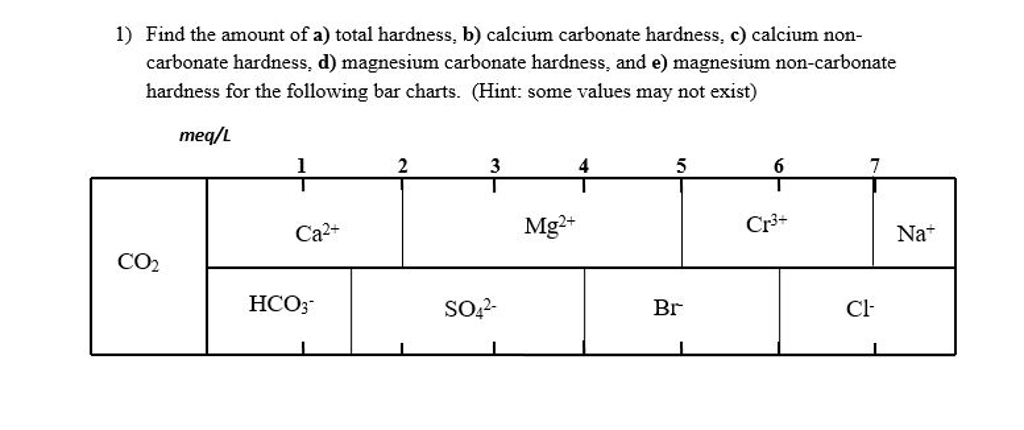
SOLVED: Problems Calculate total hardness, carbonate hardness, and non-carbonate hardness of the samples What is the degree ofhardness of the sample (sof, moderate; hard, very hard)? Why might we necd to know