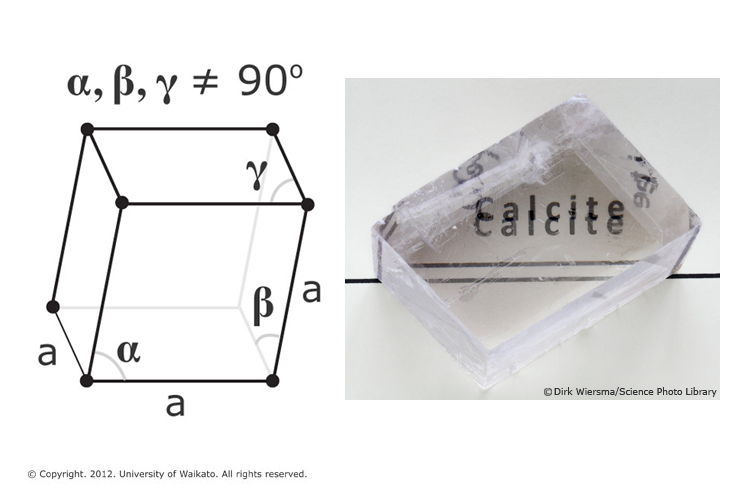
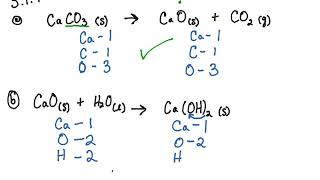Factors controlling and influencing polymorphism, morphology and size of calcium carbonate synthesized through the carbonation route: A review - ScienceDirect

Synthesis of calcium hydroxyapatite from calcium carbonate and different orthophosphate sources: A comparative study - ScienceDirect
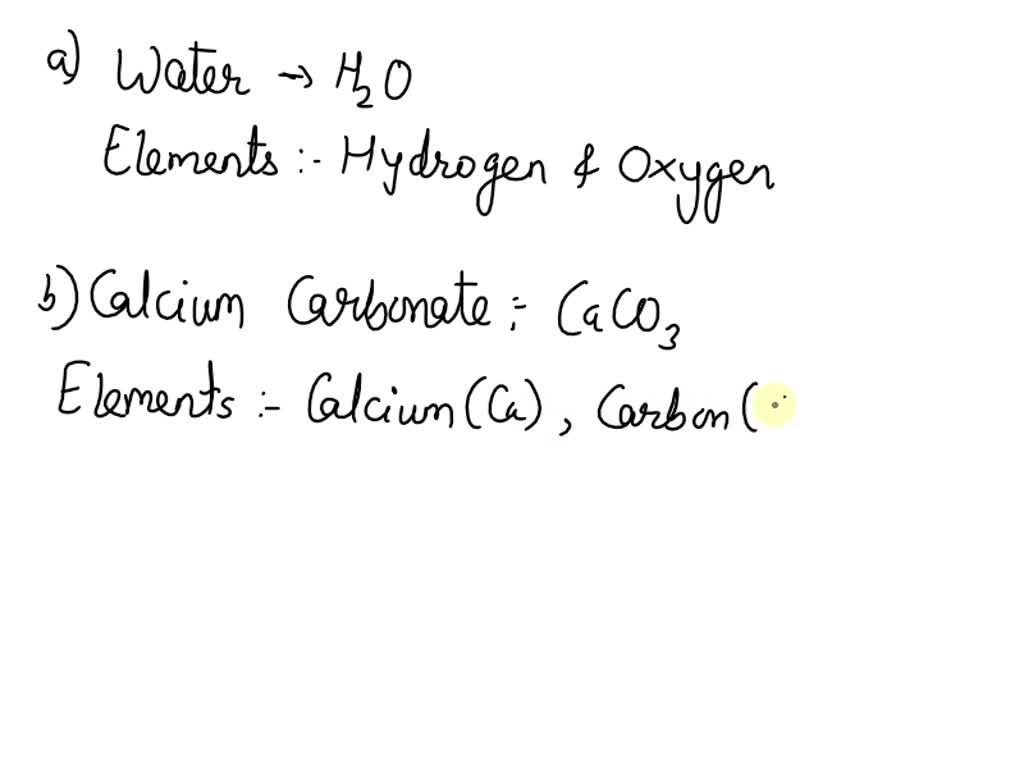
SOLVED: guide question:Q1: what are the element present in this compound: (a)water(b) calcium carbonate (c) phosphoric acid?

One-Step Synthesis of Calcium Hydroxyapatite from Calcium Carbonate and Orthophosphoric Acid under Moderate Conditions | Industrial & Engineering Chemistry Research

Urease-aided calcium carbonate mineralization for engineering applications: A review - ScienceDirect

SciELO - Brasil - Abridged acid-base wet-milling synthesis of high purity hydroyapatite Abridged acid-base wet-milling synthesis of high purity hydroyapatite
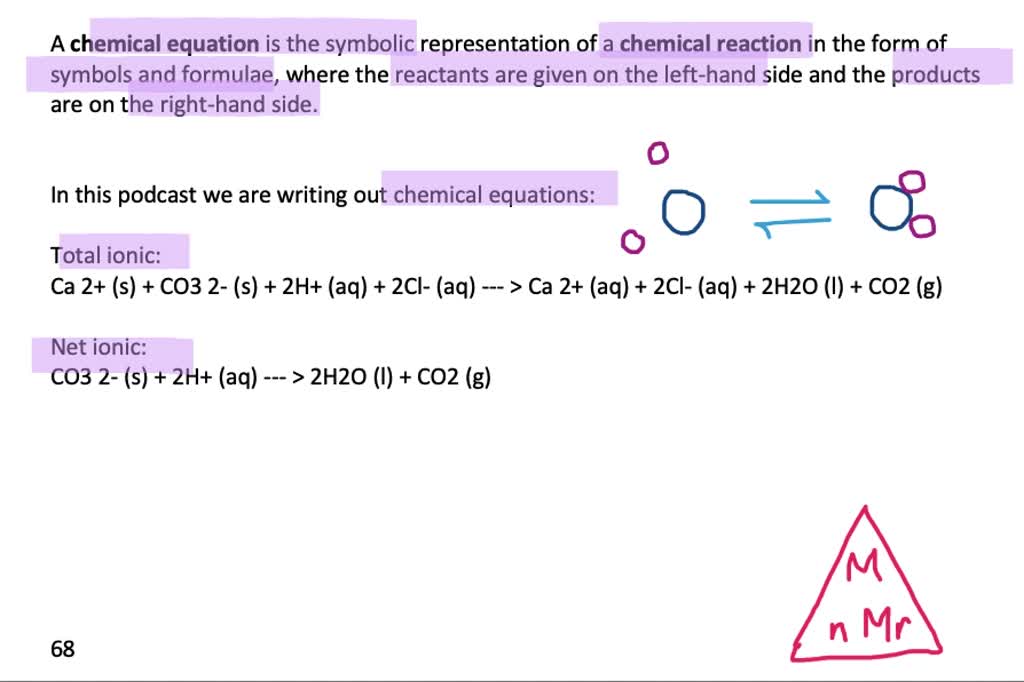
SOLVED: In the stomach, both the carbonate and phosphate ions react with the hydrochloric acid (HCl) in your stomach to become carbonic and phosphoric acid. When this happens, the calcium and chloride

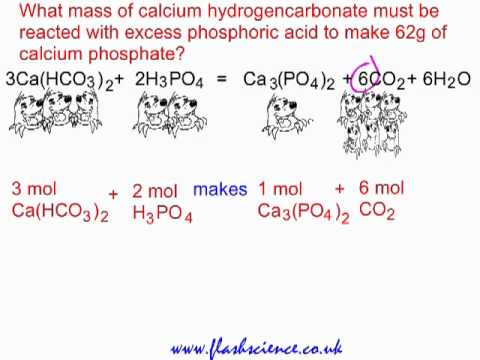
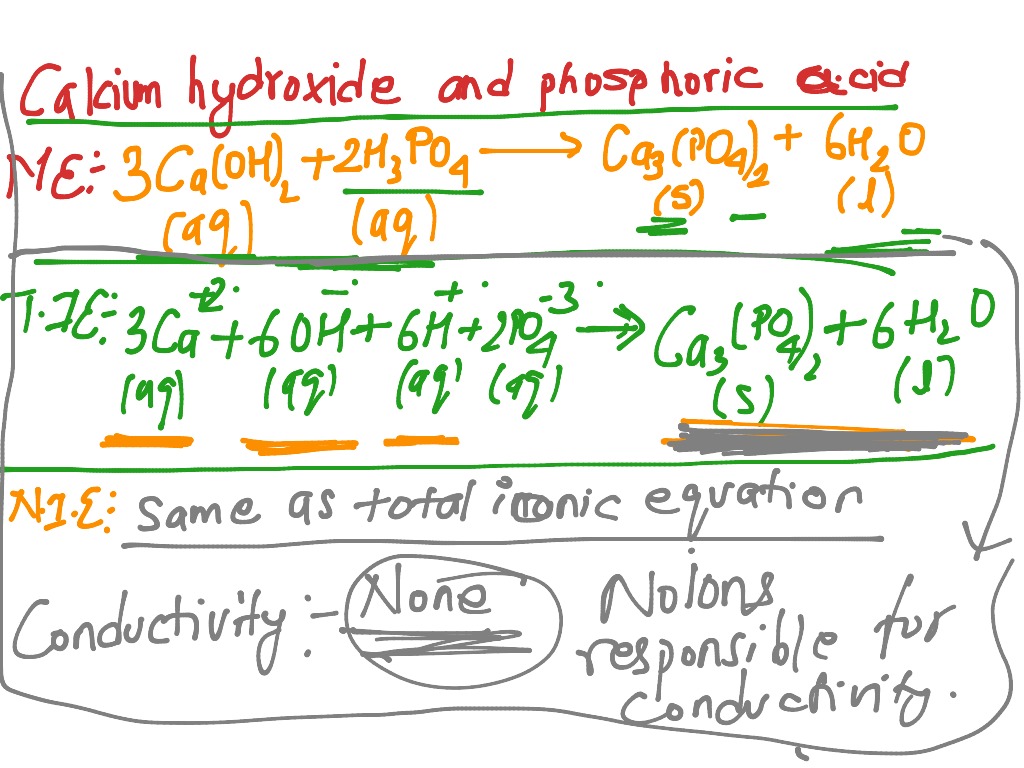
SOLVED:Write balanced chemical equations for each of the following processes: (a) Calcium phosphate reacts with sulfuric acid to produce calcium sulfate and phosphoric acid. (b) Calcium phosphate reacts with water containing dissolved