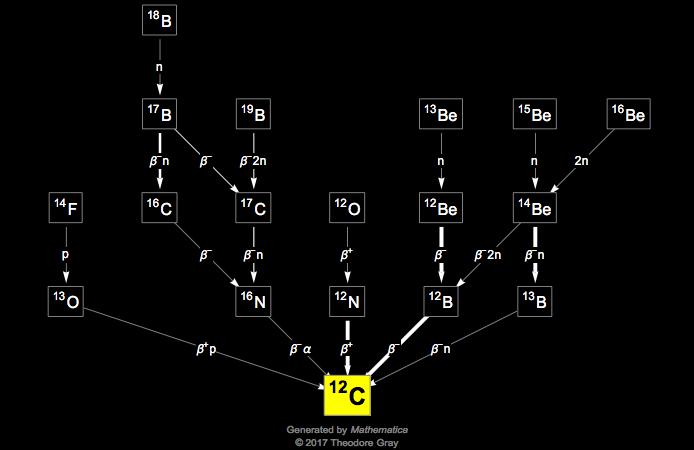
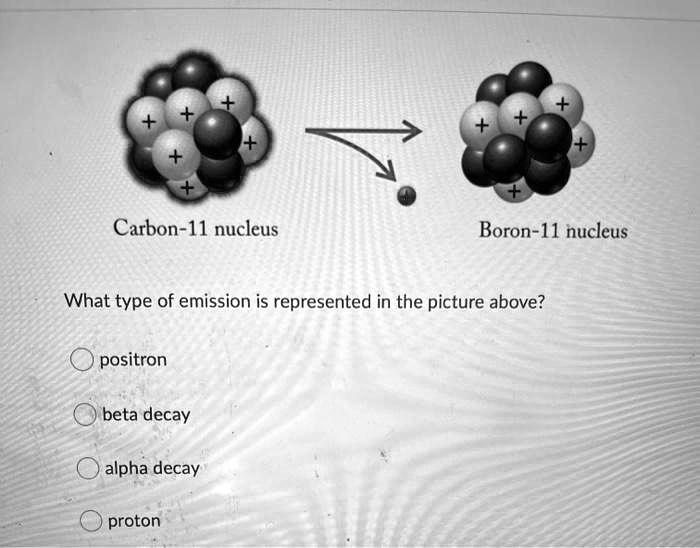
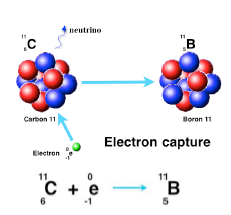
Carbon - 11 decays to boron - 11 according to the following formula. ^116C 5^11B + e^+ + ve + 0.96MeV Assume that positrons (e^+) produced in the decay combine with free

SOLVED:Carbon- 11(^116 C) undergoes beta-plus decay. Determine the product of the decay and the energy released.

SOLVED: Carbon- 11(6^11C) undergoes beta-plus decay. Determine the product of the decay and the energy released.
Carbon-II decays to boron-II according to the following formula. 11 6 C → 11 5 B + e^+ + ve + 0.96 MeV - Sarthaks eConnect | Largest Online Education Community

Carbon (Z=6) with mass number 11 decays to boron (Z=5).(a) Is it a beta^+ - decay? (b) the half-l... - YouTube
![11C]Carbon Dioxide: Starting Point for Labeling PET Radiopharmaceuticals | IntechOpen 11C]Carbon Dioxide: Starting Point for Labeling PET Radiopharmaceuticals | IntechOpen](https://www.intechopen.com/media/chapter/58065/media/F2.png)



![11C]Carbon Dioxide: Starting Point for Labeling PET Radiopharmaceuticals | IntechOpen 11C]Carbon Dioxide: Starting Point for Labeling PET Radiopharmaceuticals | IntechOpen](https://www.intechopen.com/media/chapter/58065/media/F3.png)