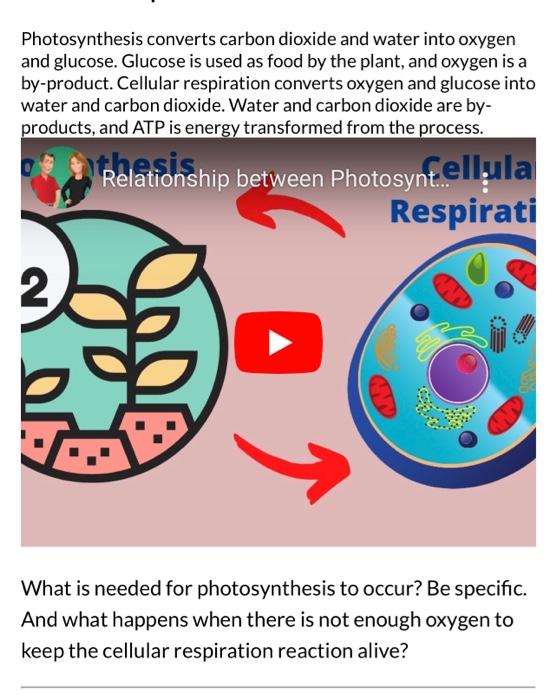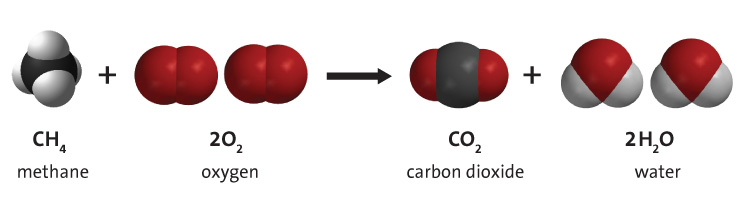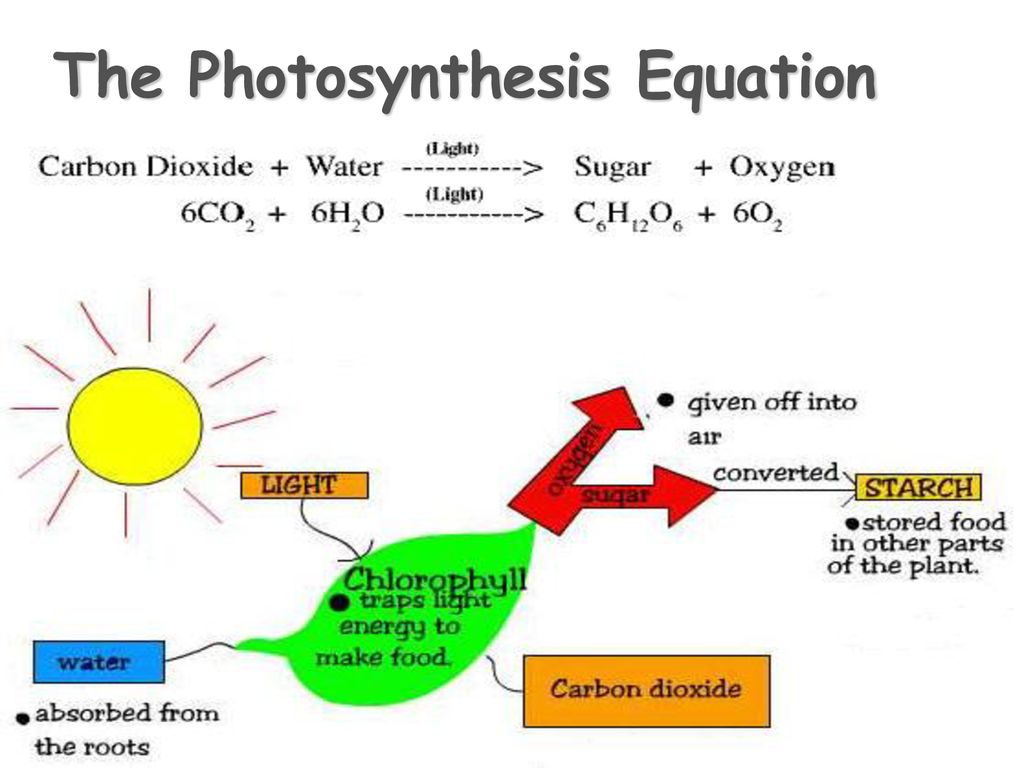
Photosynthesis Involves the Use Of light Energy to convert Water (H20) and Carbon Dioxide (CO2) into Oxygen (O2) and High Energy Carbohydrates. - ppt download

Which product is formed when carbon dioxide and water react in same ratio? - Find 4 Answers & Solutions | LearnPick Resources
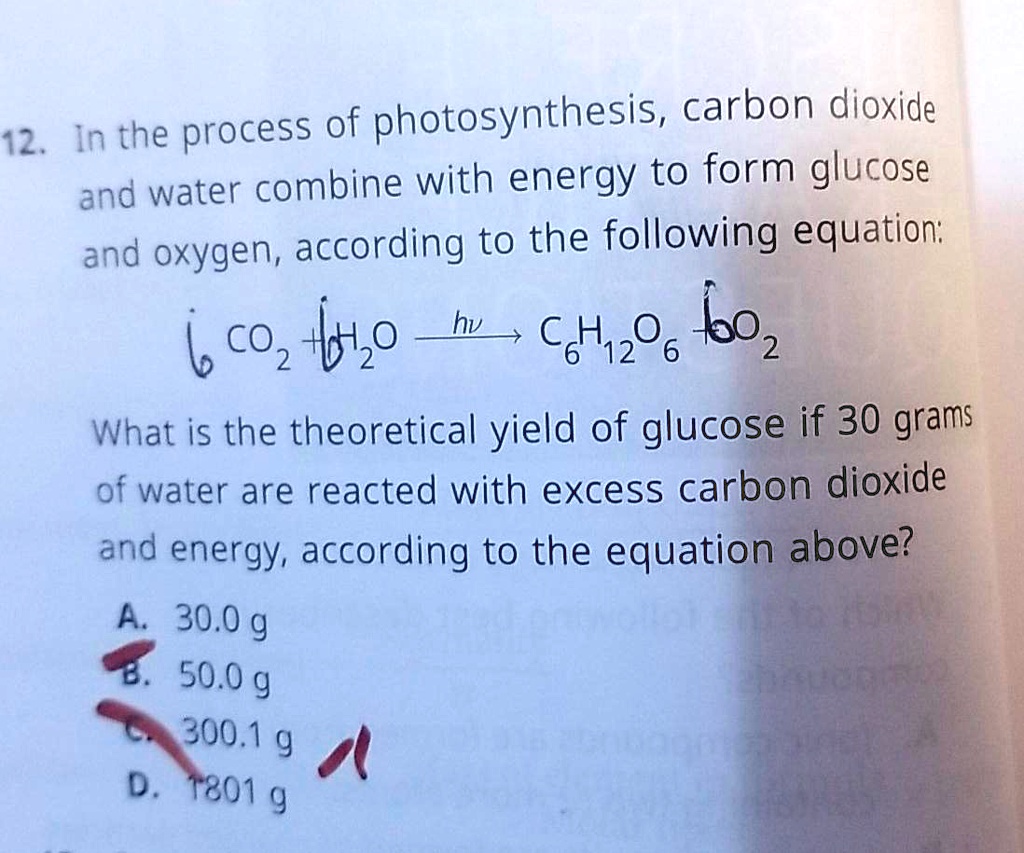
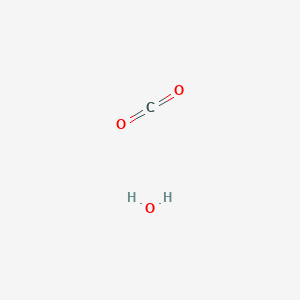
SOLVED: In the process of photosynthesis, carbon dioxide (CO2) and water (H2O) combine with energy to form glucose (C6H12O6) and oxygen (O2), according to the following equation: CO2 + H2O + energy ->

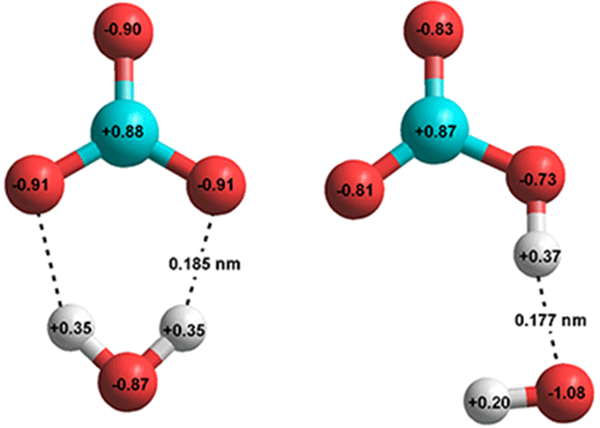
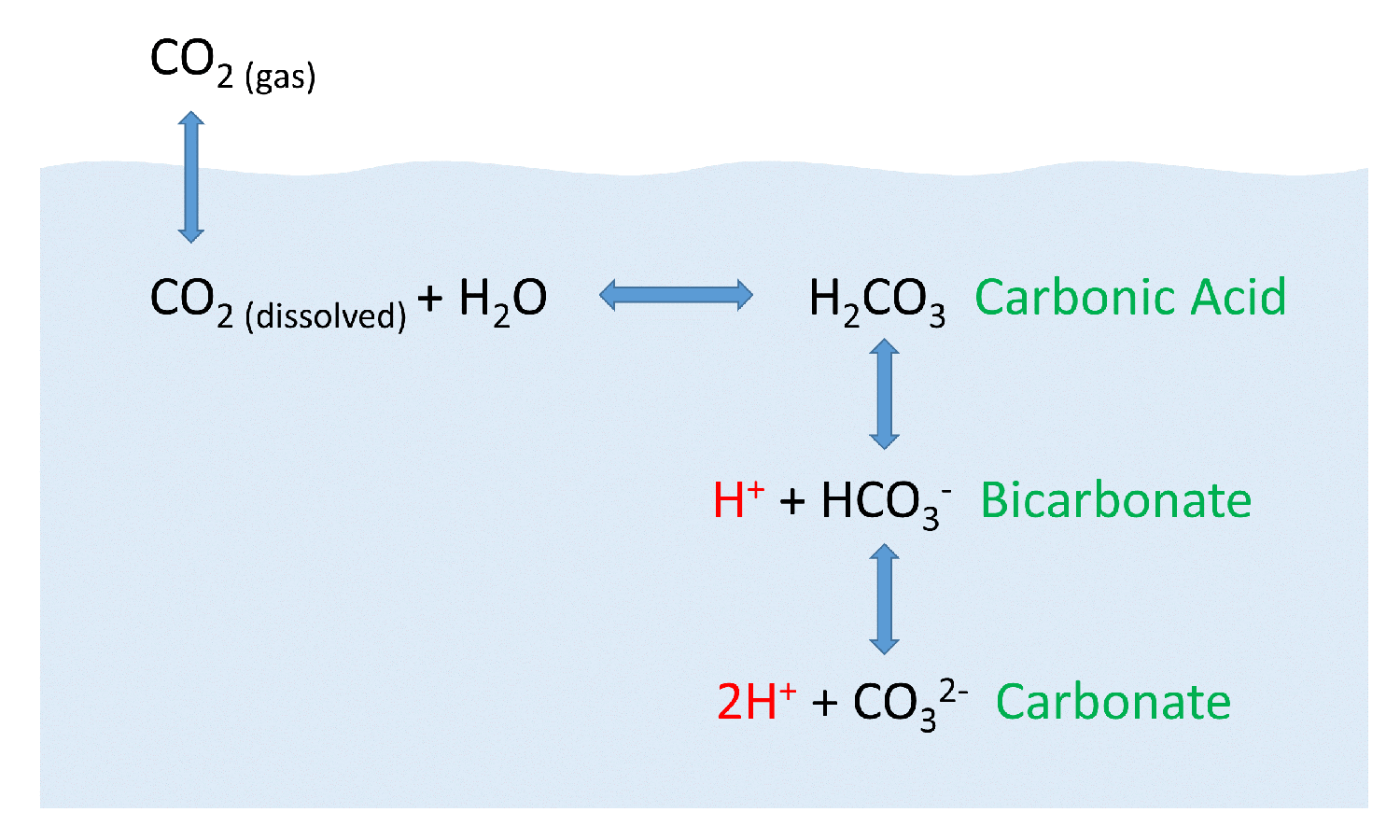
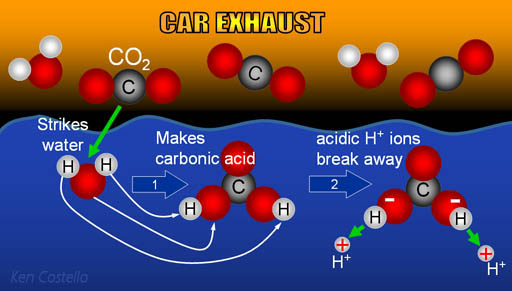
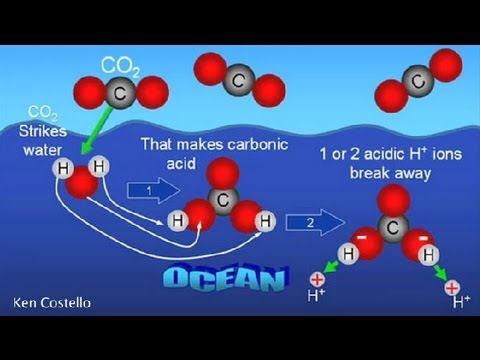
SOLVED: Carbon dioxide reacts with water in our blood to form carbonic acid. This reaction is critical in maintaining constant pH values. Water Hydrogen ion H2CO3 H+ HCO3- Carbonic Bicarbonate acid ion

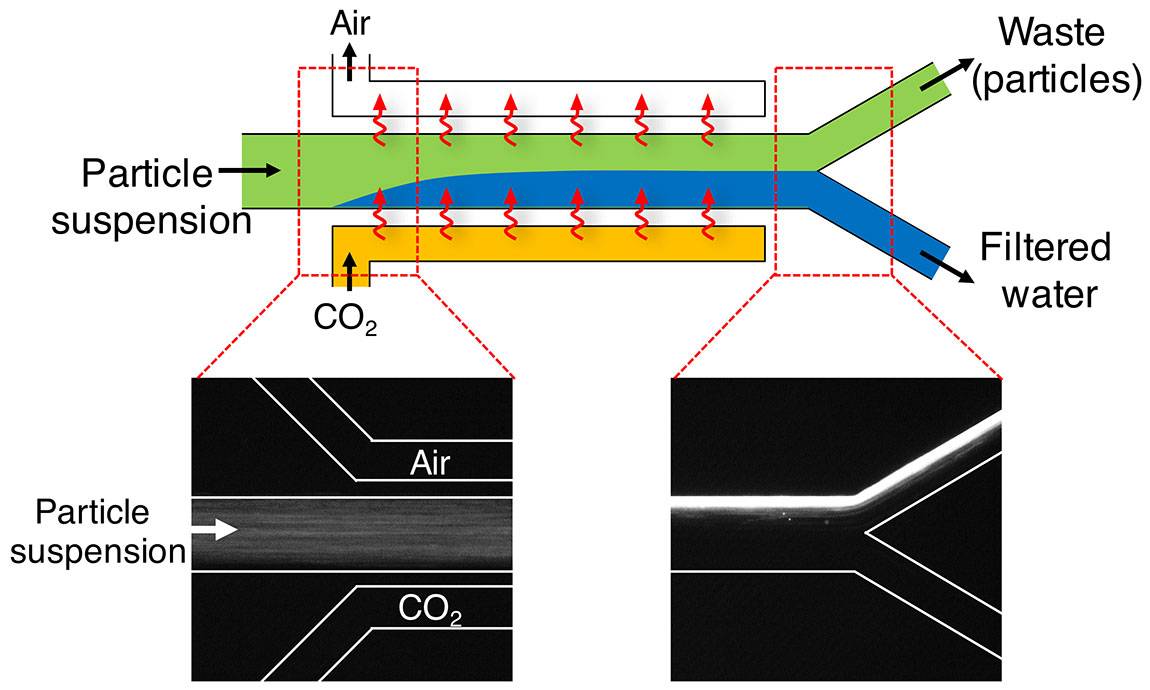
Carbon Dioxide in Water Solubility & Reaction | Is CO2 Soluble in Water? - Video & Lesson Transcript | Study.com