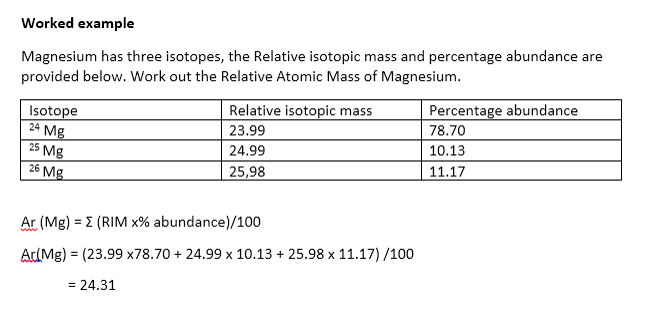
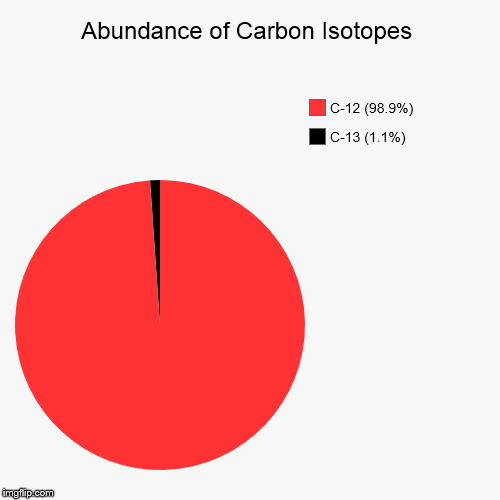
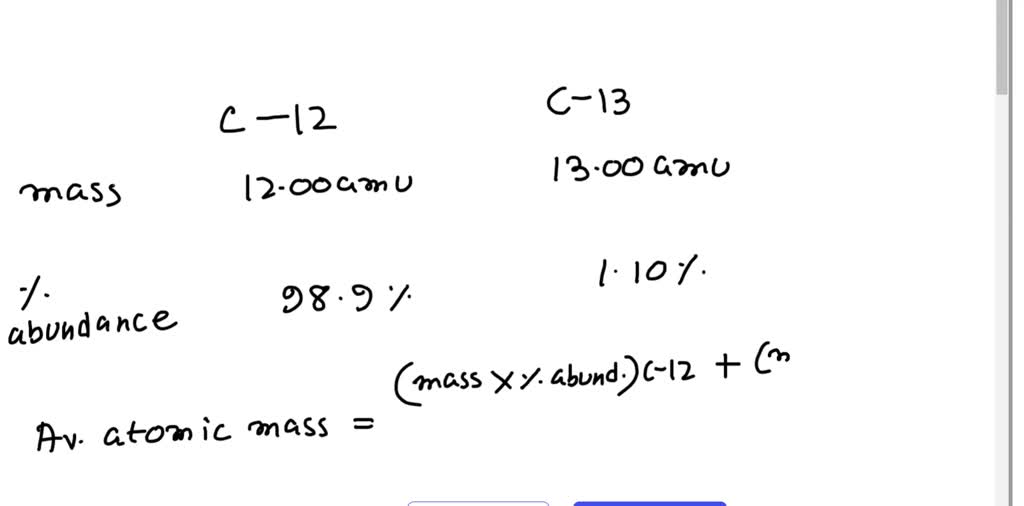The relative abundances of the stable isotopes of elements commonly... | Download Scientific Diagram
How would you calculate the relative abundance for two isotopes when the relative atomic mass is given? - Quora

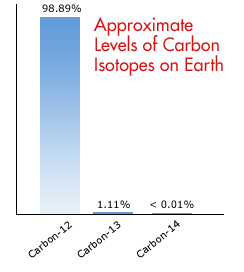
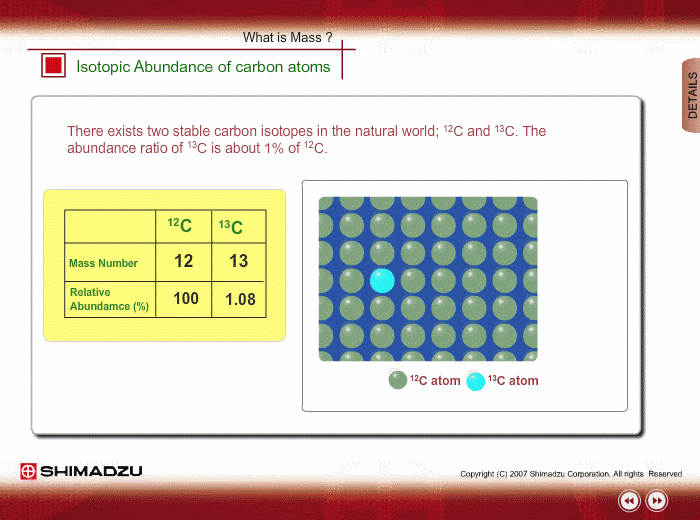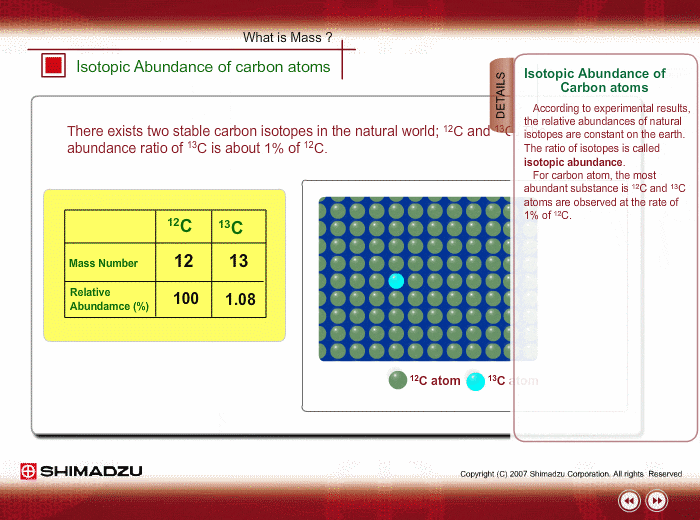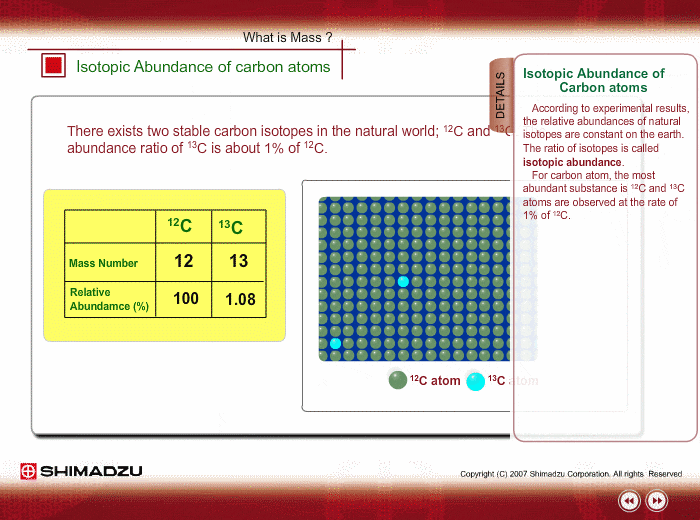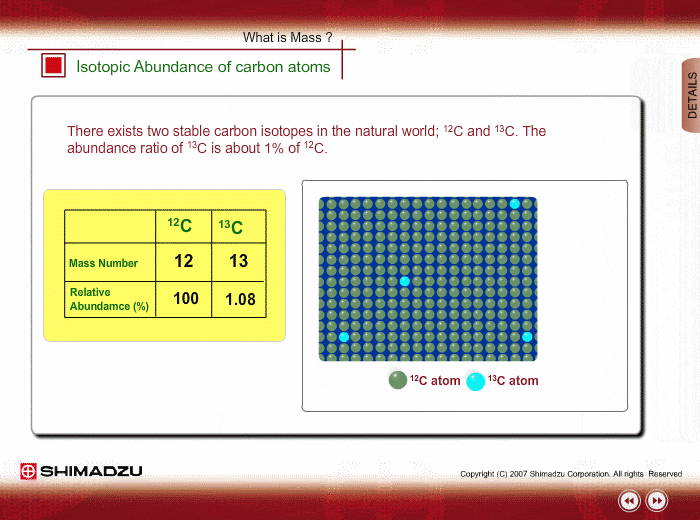
Isotopic Abundance SCH 3U. Atomic Mass The mass of an atom (protons, neutrons, electrons) Relative Atomic Mass: An element's atomic mass relative to the. - ppt download