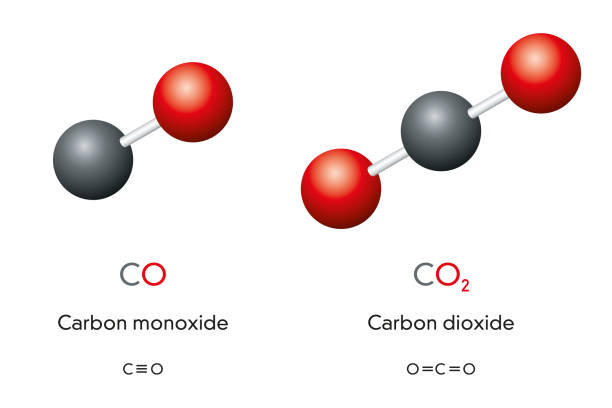
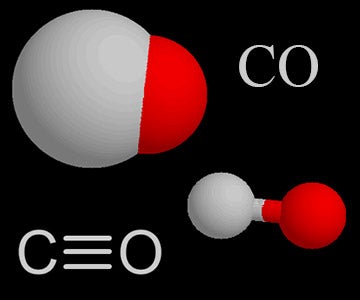
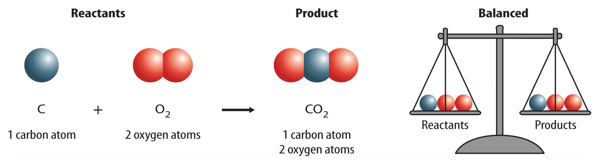
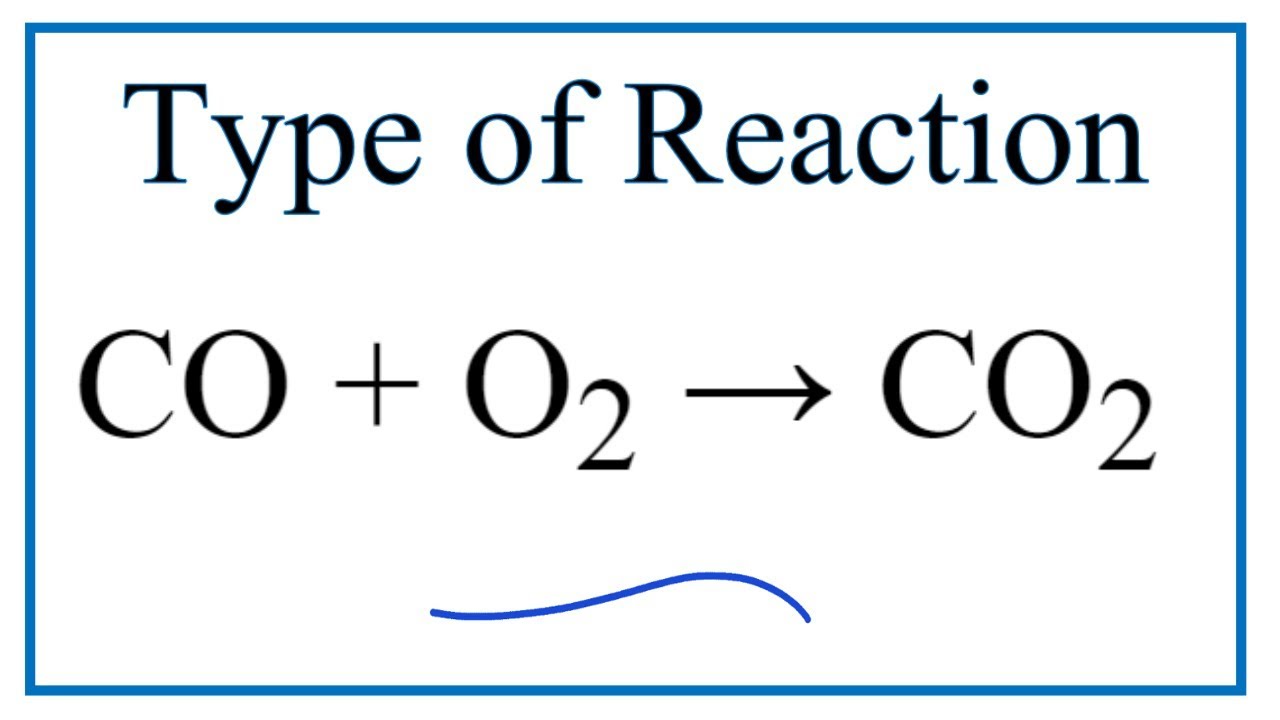write the balanced chemical equation of carbon react with oxygen when it's suppy is insufficient to form - Brainly.in

The heat of the combustion of graphite and carbon monoxide respectively are 393.5 kJ mol1 and 283 kJ mol ^-1 . Thus heat of formation of carbon monoxide in kJ mol ^-1 is:

Explosion limits of the dry carbon monoxide + oxygen reaction - Transactions of the Faraday Society (RSC Publishing)
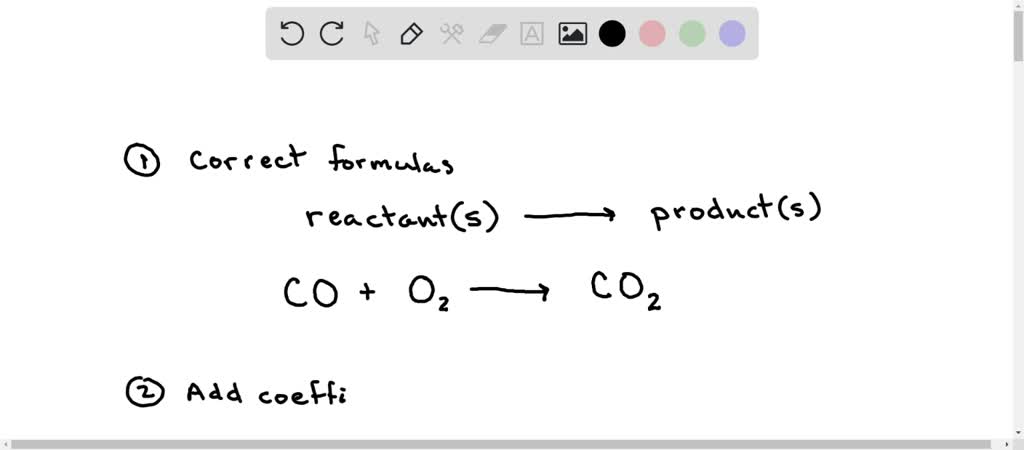
SOLVED: Write a balanced equation for the following reaction: When carbon monoxide combines with oxygen, carbon dioxide is formed. (Use the lowest possible coefficients. Omit states of matter.) Reactants Products + Step
43. Methane reacts with oxygen to produce carbon dioxide and water. a) Find out the mole reaction between reactants and products. b) Volume relation at STP between reactants and products. c) Mass
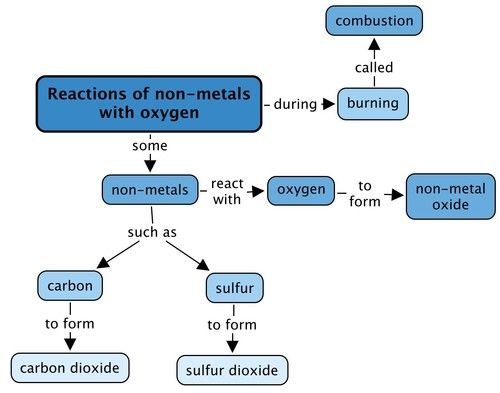
10.1 The general reaction of non-metals with oxygen | Reactions of non-metals with oxygen | Siyavula
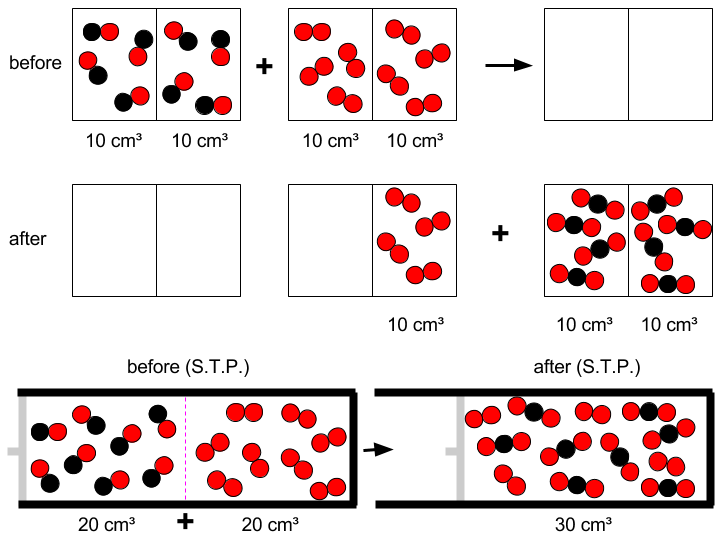
20*cm^3 volumes EACH of carbon monoxide and oxygen gas are combined and combusted. What volume of gas remains AFTER the reaction? | Socratic

SOLVED: 19. Carbon monoxide reacts with oxygen according to the equation below: 2 cO(g) + Oz(g) v 2 COz(g) What is the overall order of the reaction? a. first-order b second-order third-order
.png)