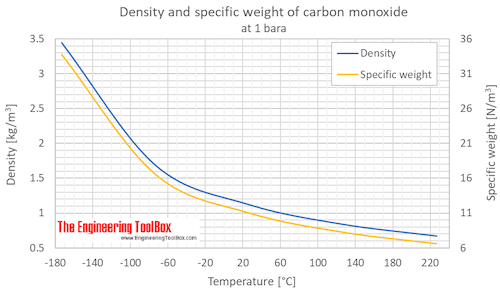
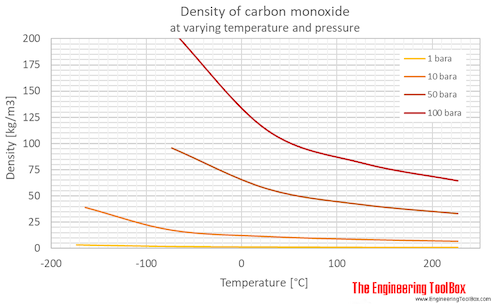
SOLVED: What is the density of carbon monoxide gas at STP? STP: Standard Temperature and Pressure (T= 0 %C , Pz 1 atm) Select one: A. 1.25 g/L B 1.96 g/L C

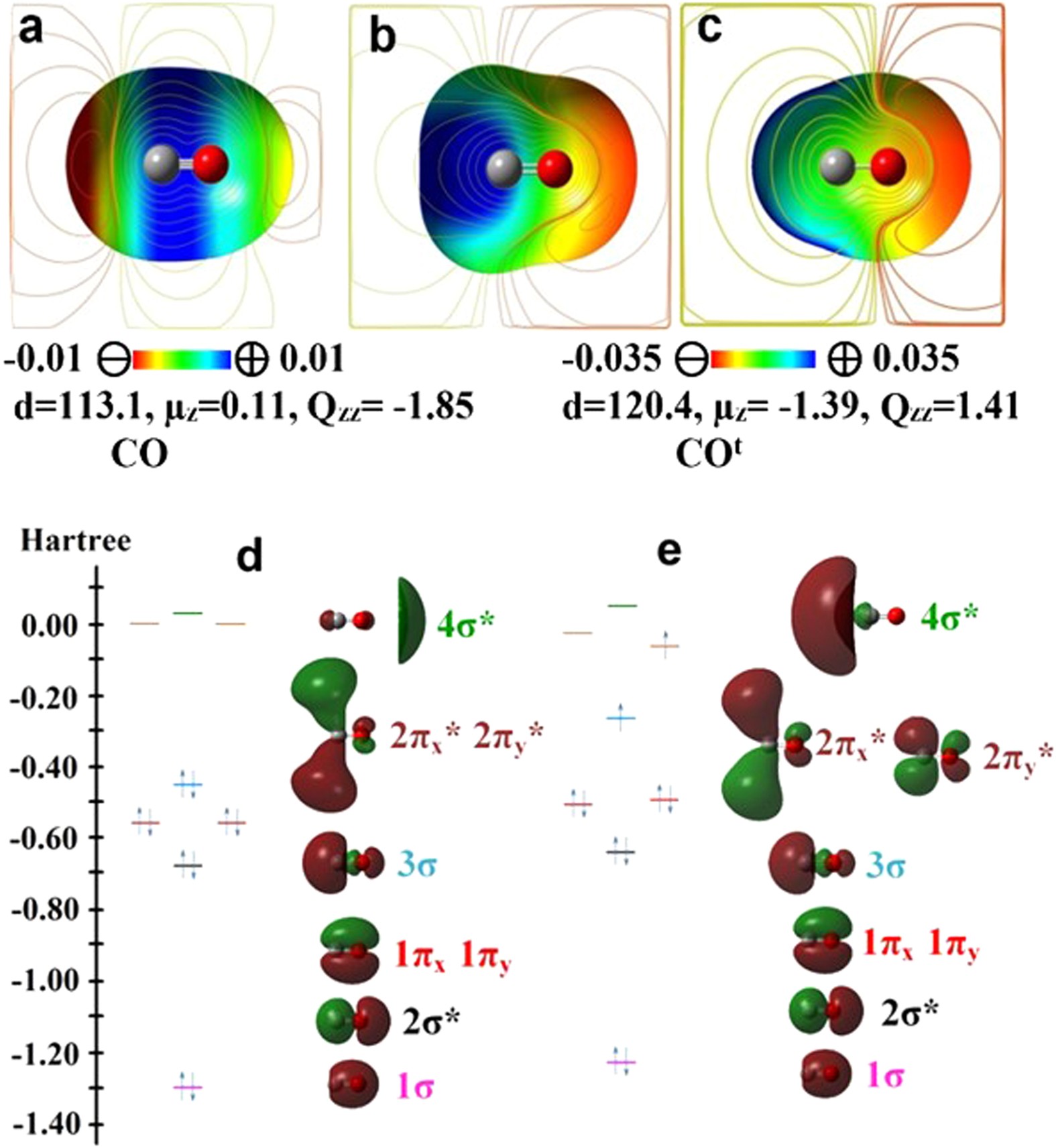
The partial density of states (PDOS) for a single CO molecule and the... | Download Scientific Diagram

Elements of modern chemistry . ions to carbon dioxide. 2 volumes CO absorb 2 volumes of chlorine to form 2 volumes CO.CP2 volumes CO absorb 1 volume of oxygen to form

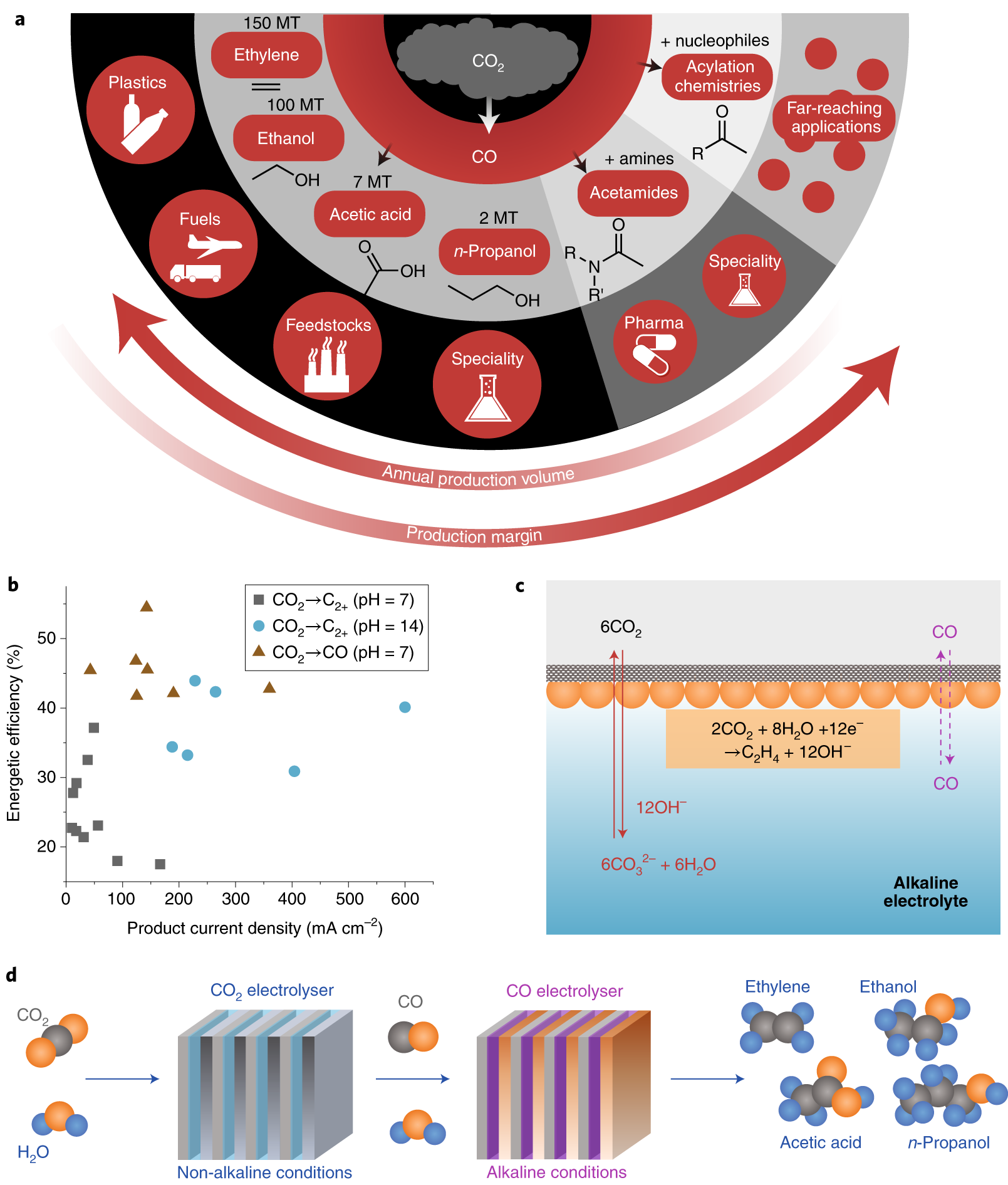
Catalysts | Free Full-Text | Density Functional Theory Study of the Hydrogenation of Carbon Monoxide over the Co (001) Surface: Implications for the Fischer–Tropsch Process

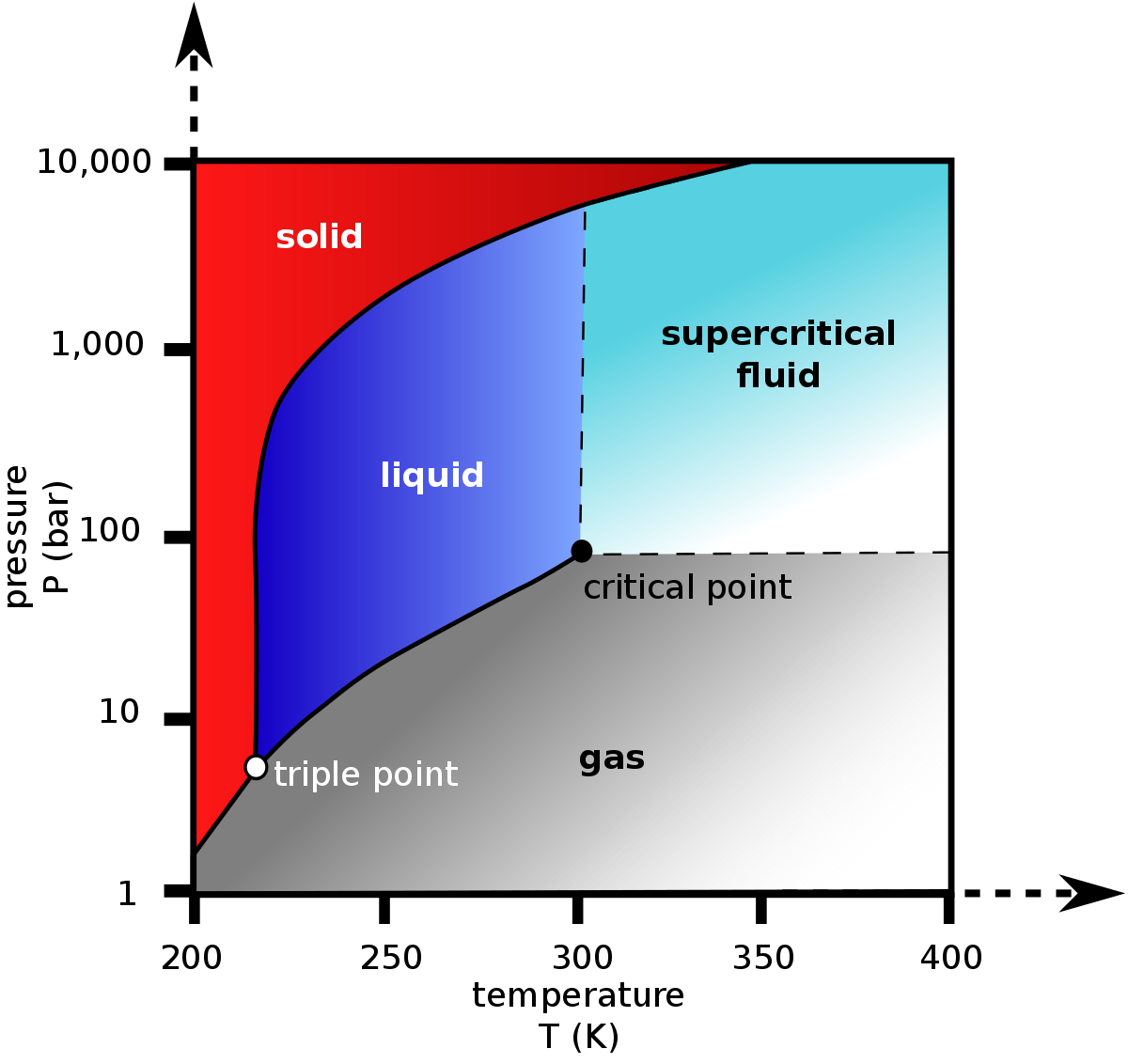
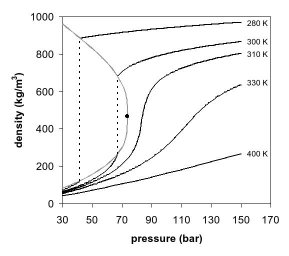
High-pressure temperature vs density diagram for carbon dioxide. Solid... | Download Scientific Diagram

Density (kg/m3) of Carbon Dioxide as a Function of Temperature and Pressure - Carbon Dioxide Thermodynamic Properties Handbook - Wiley Online Library