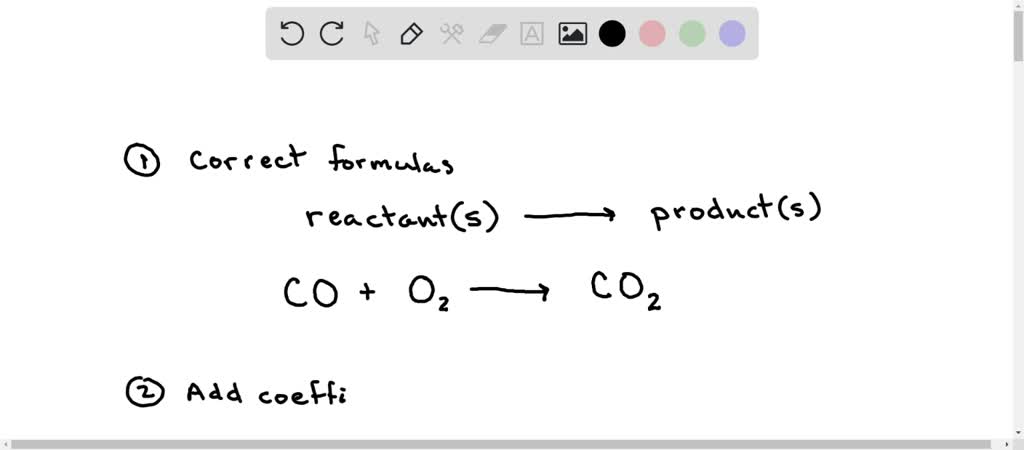
SOLVED: Write a balanced equation for the following reaction: When carbon monoxide combines with oxygen, carbon dioxide is formed. (Use the lowest possible coefficients. Omit states of matter.) Reactants Products + Step
Formation of carbon monoxide and carbon dioxide molecules: (1) empty... | Download Scientific Diagram
![SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism](https://cdn.numerade.com/ask_images/736b473b932d4d048614f30fae3d5333.jpg)
SOLVED: The rate law for the formation of carbon dioxide from carbon monoxide and oxygen is given by: 2CO(g) + 0,(g) 2C0,(g) Rate = k [CO][02] Which one of the following mechanism
62.The Enthalpies of combustion of carbon and carbon monoxide are 390 kJ and 278kJ respectively. The enthalpy of formation of carbon monooxide is? a) 669 kJ b) 112 kJ c) 112 kJ d) 668 kJ

The heats of combustion of carbon and carbon monoxide are - 393.5 and - 283.5 kJ mol ^-1 , respectively. The heat of formation (in kJ) of carbon monoxide per mole is:
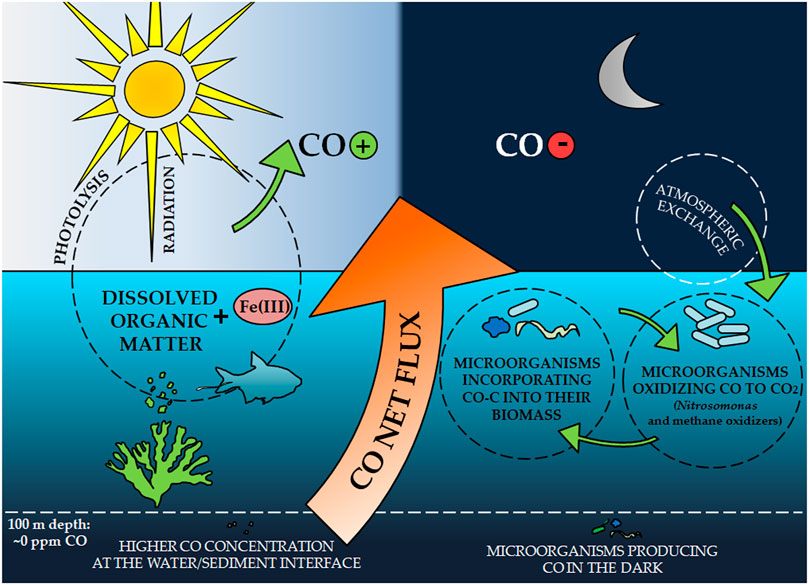
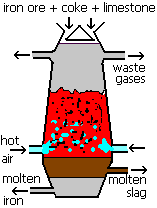
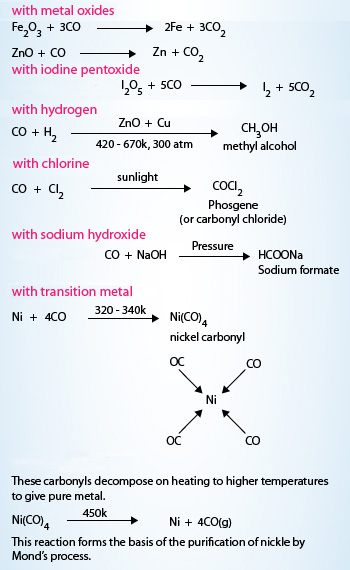
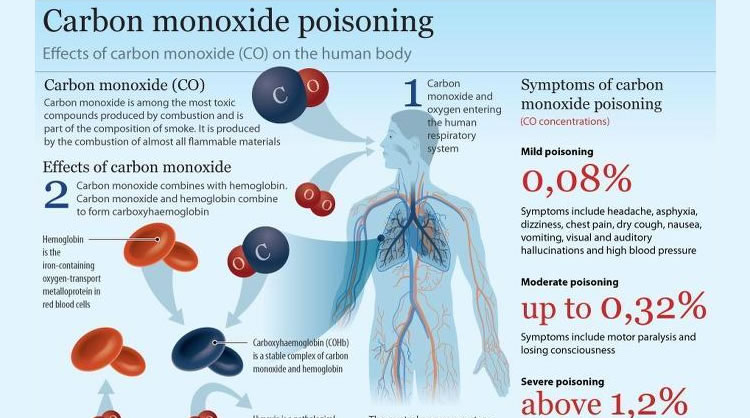
Frontiers | Carbon Monoxide Fate in the Environment as an Inspiration For Biorefinery Industry: A Review

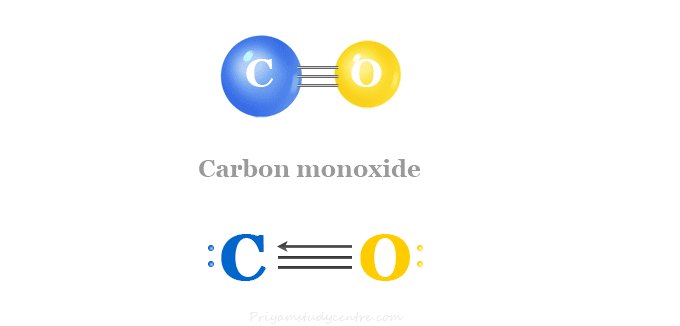

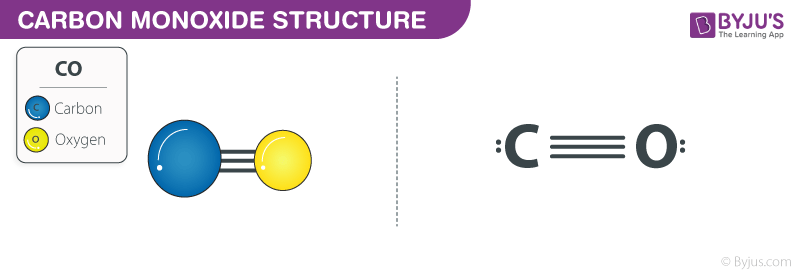
:max_bytes(150000):strip_icc()/what-is-carbon-monoxide-Final-27d97041ac95424c9af73c0dd25adcad.jpg)