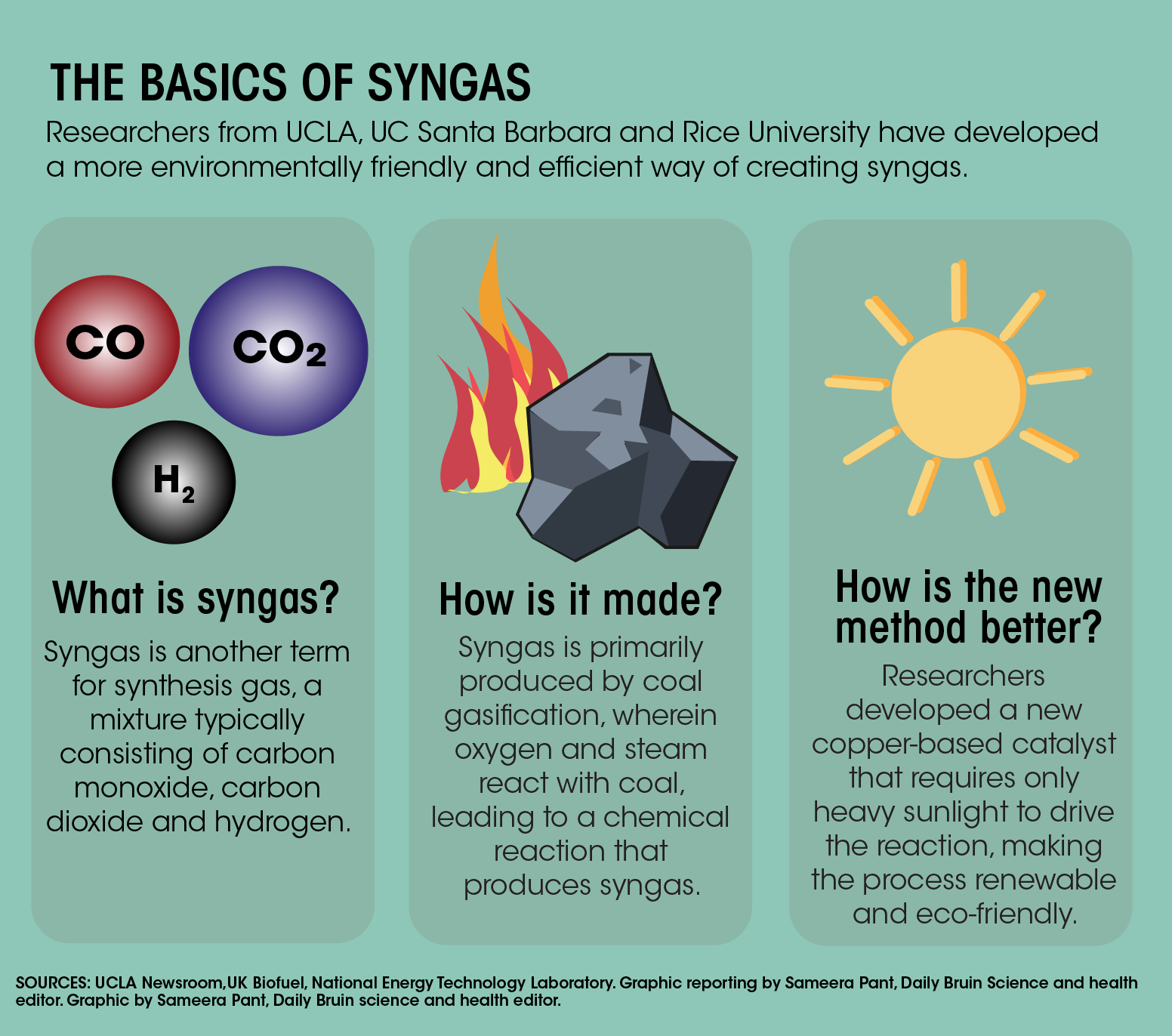
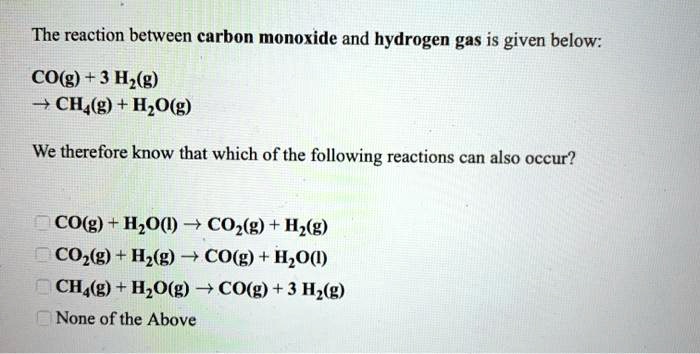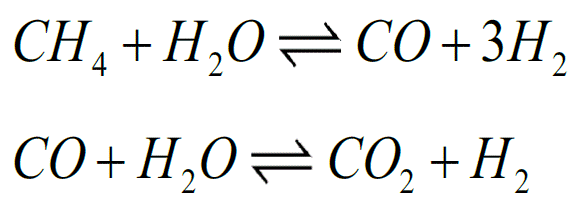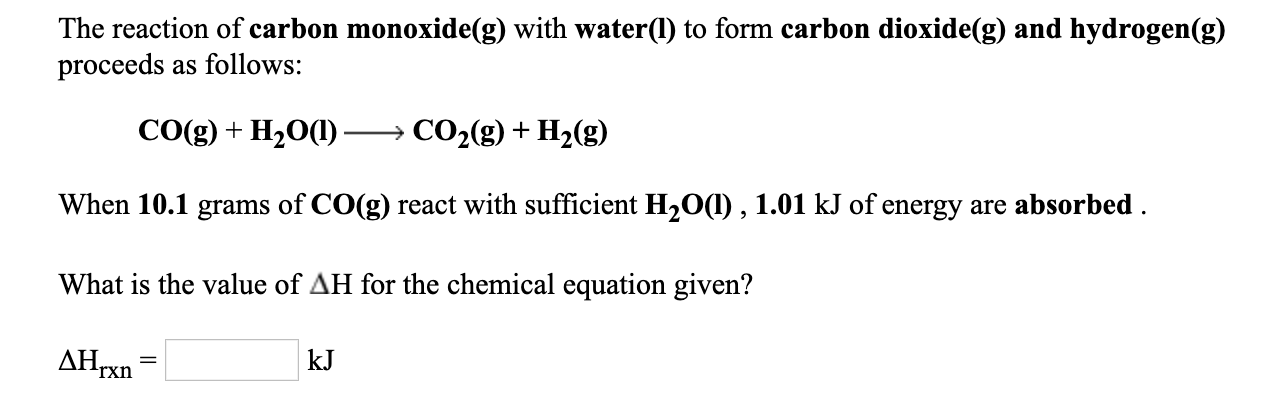carbon monoxide reacts with hydrogen under certain condition to form methanol (CH3OH). write the balanced - Brainly.in

Propene reacts with carbon monoxide and hydrogen in presence of cobalt carbonyl catalyst at high - YouTube
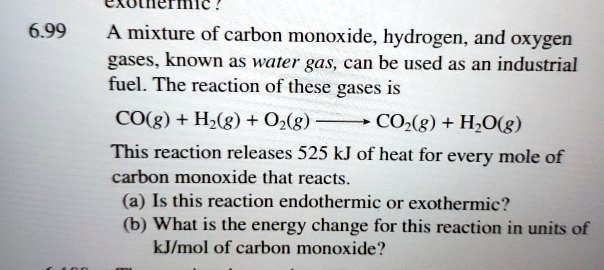
SOLVED: CAOICMIC 6.99 A mixture of carbon monoxide, hydrogen, and oxygen gases; known as water gas; can be used as an industrial fuel. The reaction of these gases CO(g) + Hz(g) +

Natural gas consists primarily of methane, CH 4.It is used in a process called stream reforming to prepare a gaseous mixture of carbon monoxide and hydrogen. - ppt download

Carbon Monoxide as a Promoter of Atomically Dispersed Platinum Catalyst in Electrochemical Hydrogen Evolution Reaction | Journal of the American Chemical Society
The Equilibrium between Carbon Monoxide, Hydrogen, Formaldehyde and Methanol.1 I. The Reactions CO + H2 ⥨ HCOH and H2 + HCOH ⥦ CH3OH | Journal of the American Chemical Society

carbon monoxide reacts with hydrogen under certain conditions to form Methanol write a balanced chemical - Brainly.in
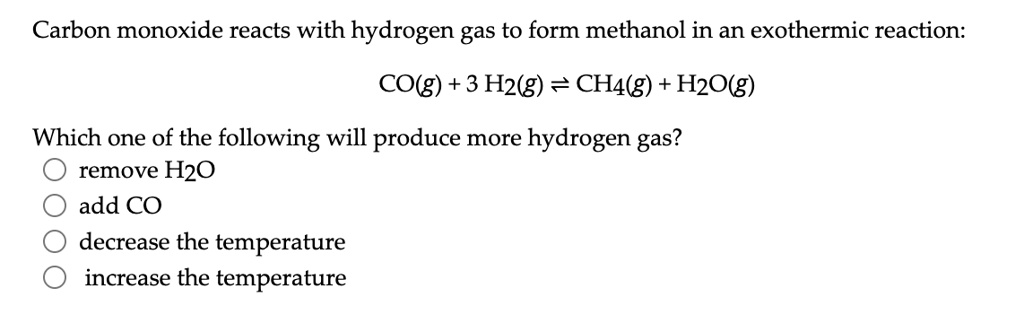
SOLVED: Carbon monoxide reacts with hydrogen gas to form methanol in an exothermic reaction: CO(g) 3 Hz(g) = CH4(g) + H2Okg) Which one of the following will produce more hydrogen gas? remove
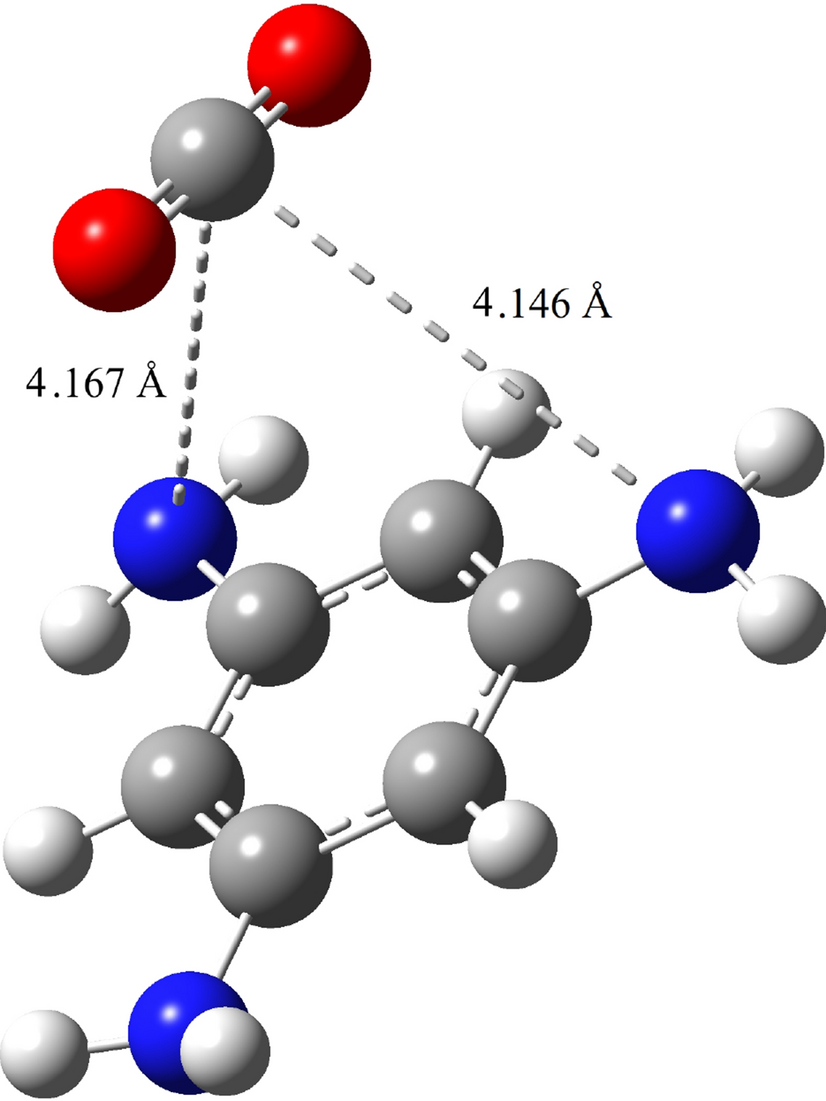
A new approach to separate hydrogen from carbon dioxide using graphdiyne-like membrane | Scientific Reports