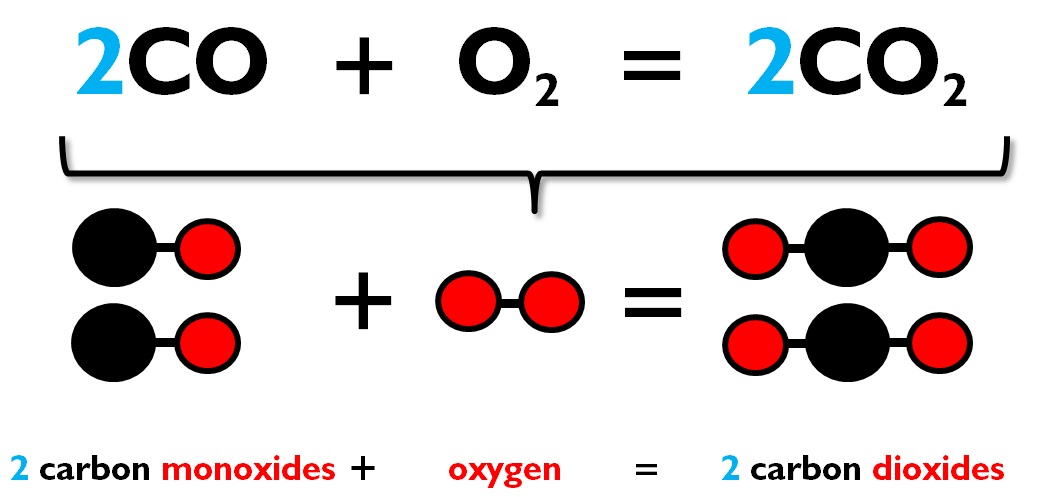
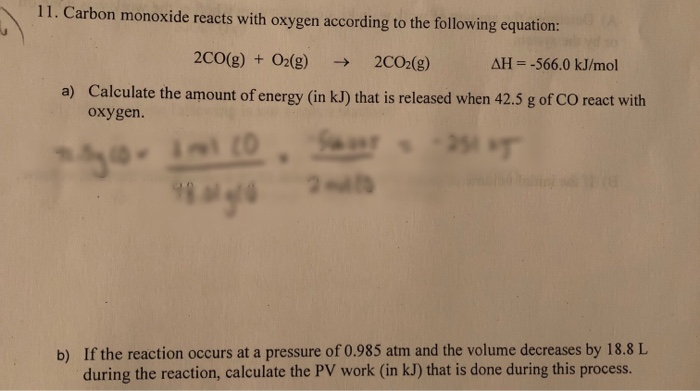Carbon disulfide reacts with oxygen gas to produce carbon dioxide and sulfur dioxide, 1) Write the balanced - Brainly.com

3g of Carbon (C) reacts completely with 5g of Oxygen (O2). Calculate the amount of Carbon monoxide (CO) produced.
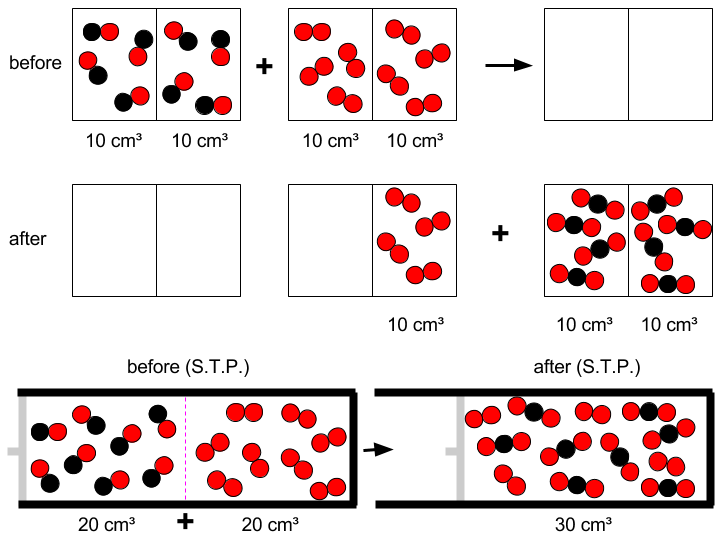
20*cm^3 volumes EACH of carbon monoxide and oxygen gas are combined and combusted. What volume of gas remains AFTER the reaction? | Socratic
.png)
how will carbon react with oxygen what happens when the product is dissolved in water show along with - Science - Materials Metals and Non-Metals - 1217495 | Meritnation.com

SOLVED: 19. Carbon monoxide reacts with oxygen according to the equation below: 2 cO(g) + Oz(g) v 2 COz(g) What is the overall order of the reaction? a. first-order b second-order third-order

write the balanced chemical equation of carbon react with oxygen when it's suppy is insufficient to form - Brainly.in
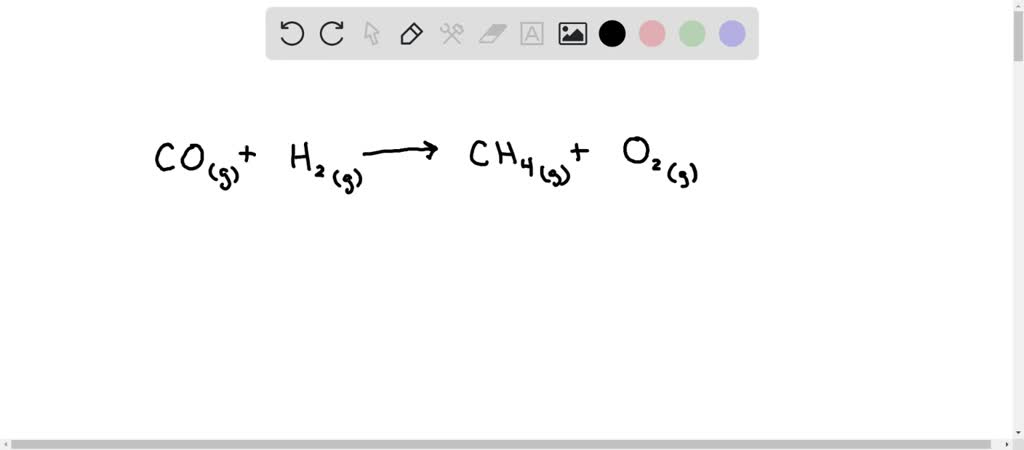
SOLVED: Gaseous carbon monoxide reacts with hydrogen gas to form gaseous methane (CH4) and oxygen gas.

SOLVED: 'Carbon monoxide reacts with oxygen to produce carbon dioxide If 1.0 L of carbon monoxide reacts with oxygen at STP conditions how many liters of oxygen are required to completely react,