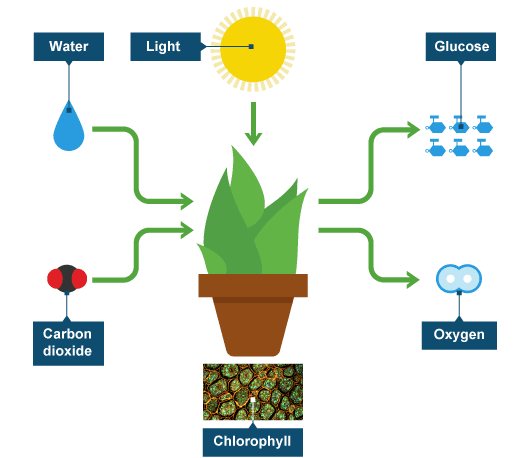
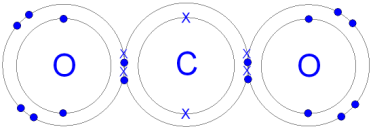
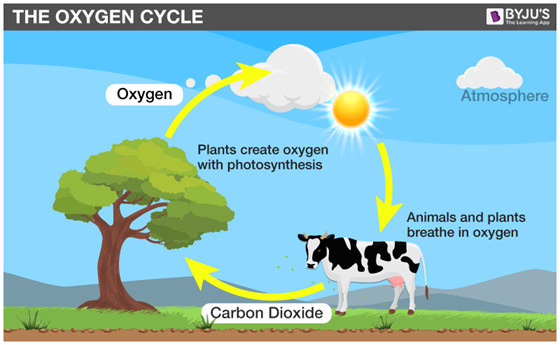
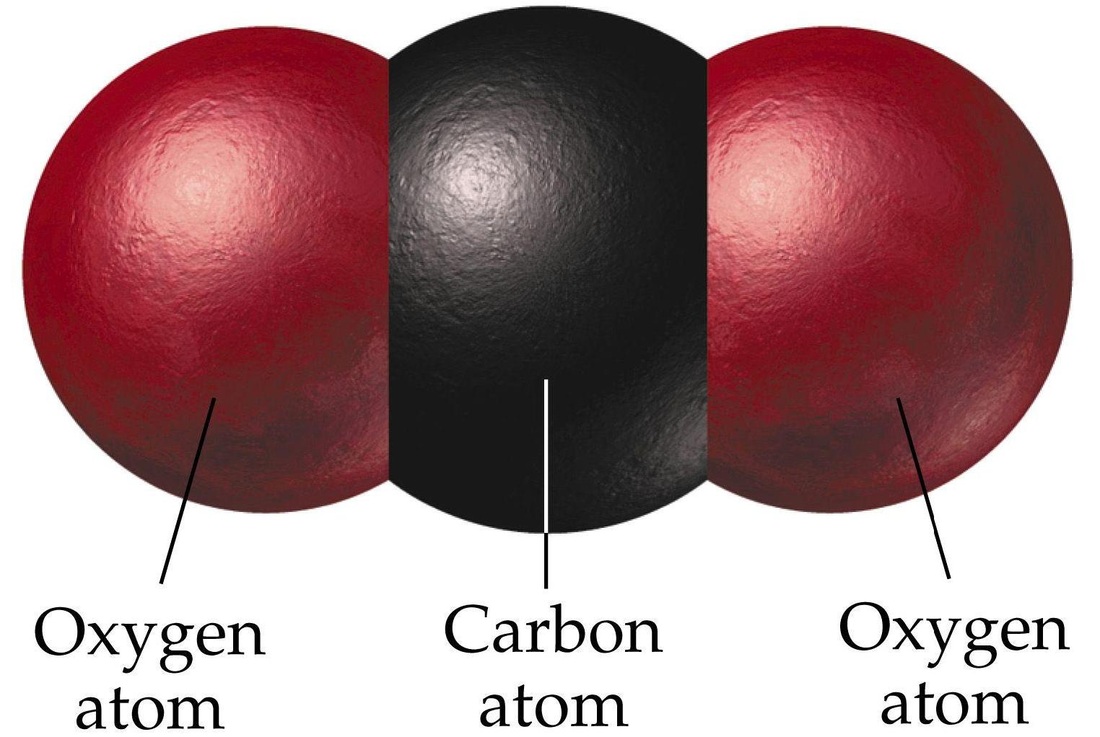
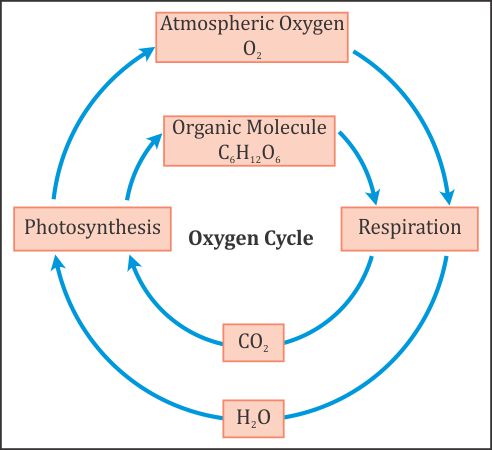
.png)
how will carbon react with oxygen what happens when the product is dissolved in water show along with - Science - Materials Metals and Non-Metals - 1217495 | Meritnation.com

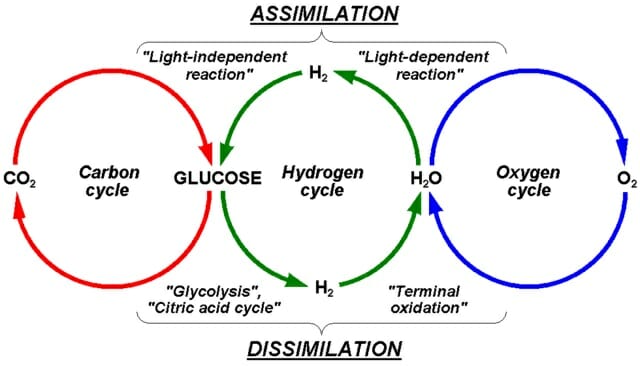
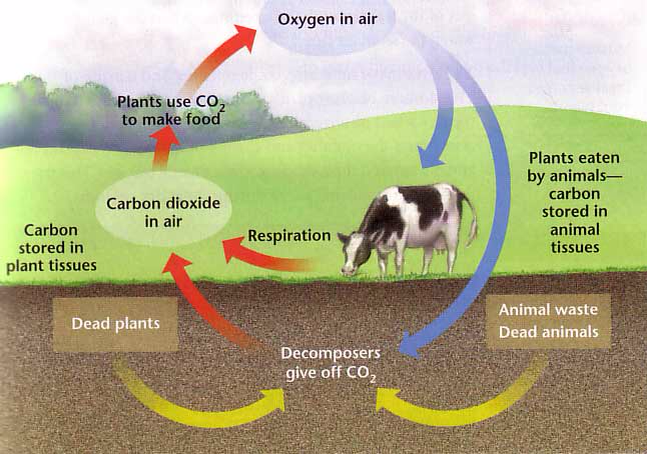
Label and describe the processes/events involve in the carbon-oxygen cycle•Atmospheric Carbon - Brainly.ph

Archivo:Atmosphere representation - nitrogen oxygen carbon dioxide water molecules.svg - Wikipedia, la enciclopedia libre

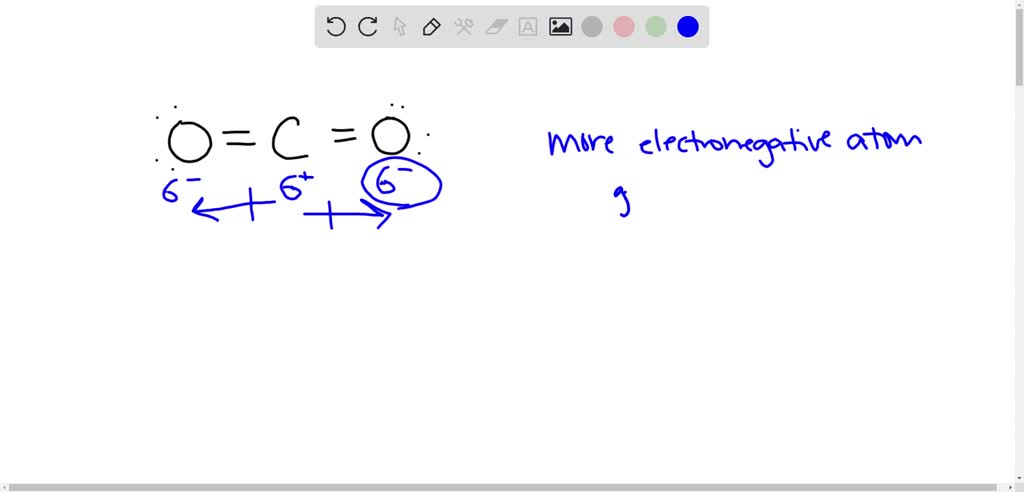
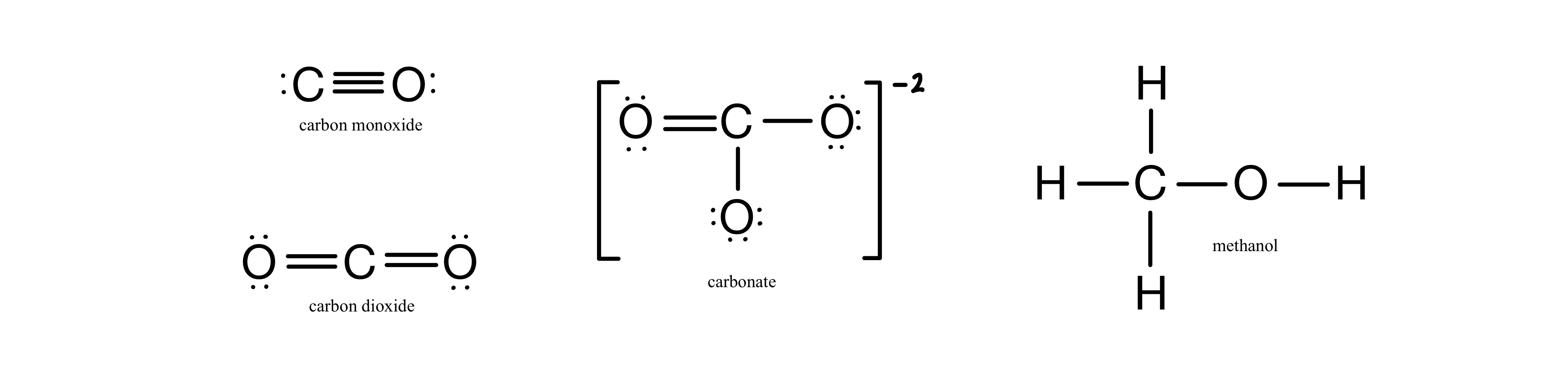
Order the following species with respect to the carbon-oxygen bond length (longest to shortest): CO, CO_2, CO_3^{2-}, CH_3OH What is the order from the weakest to the strongest carbon-oxygen bond? | Homework.Study.com
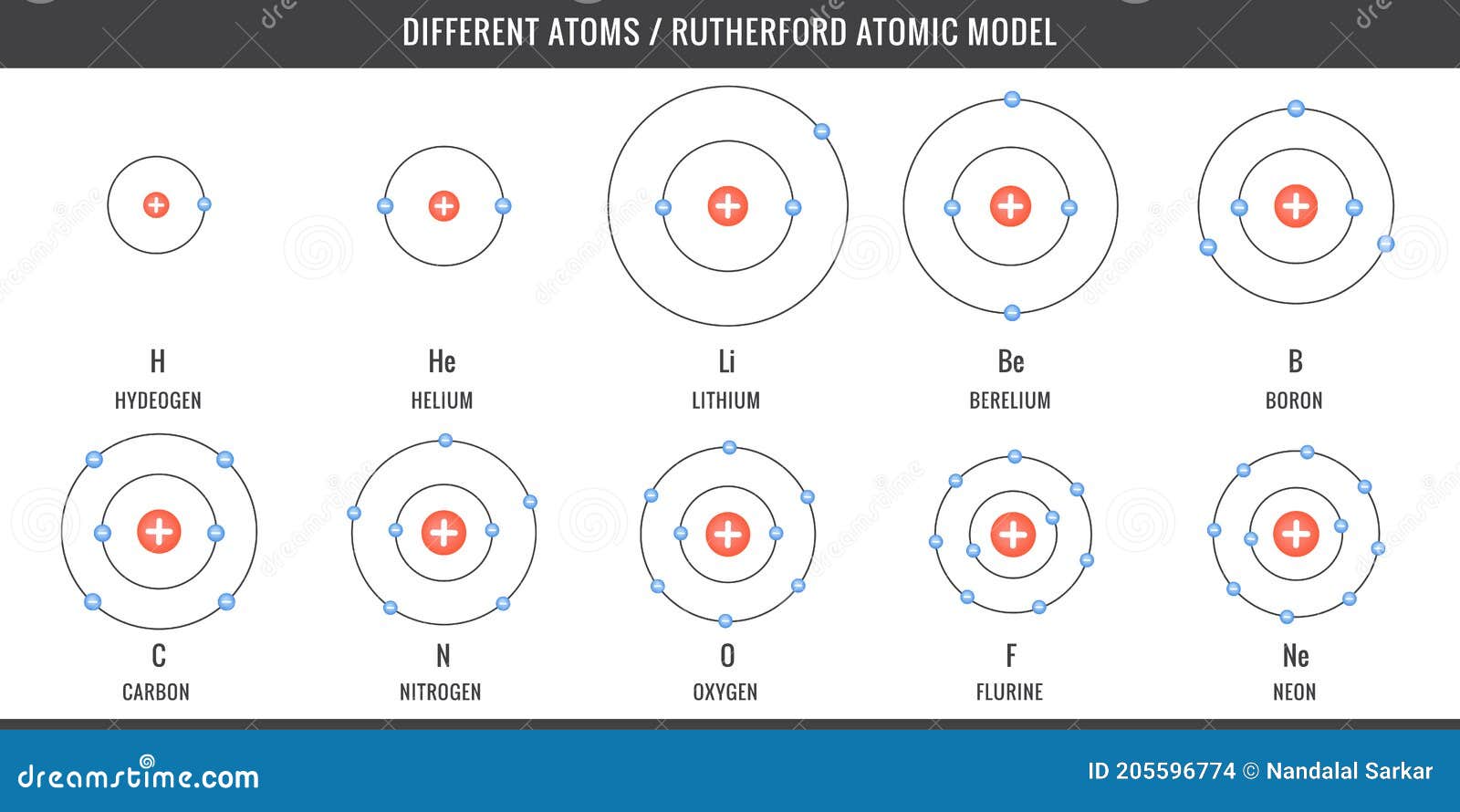
Different Atoms, Simplest Atomic Model of Hydrogen, Carbon, Oxygen, Nitrogen Stock Vector - Illustration of diagram, atom: 205596774
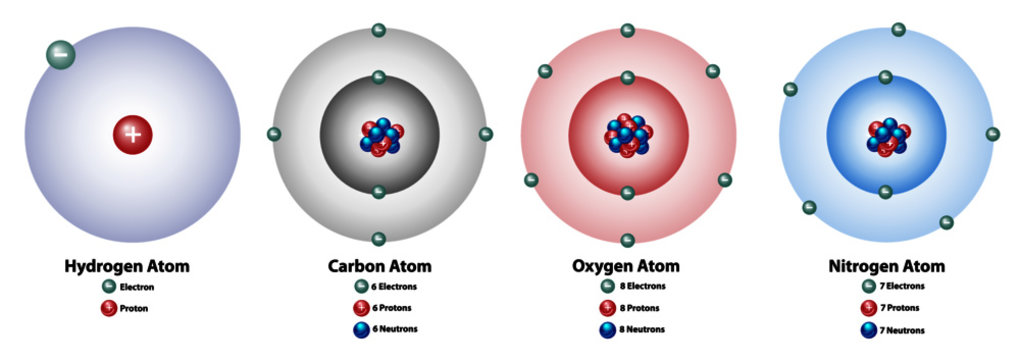
Atomic elements showing the nucleus and shells, numbers of electrons, protons, and neutrons. Hydrogen, carbon, oxygen, and nitrogen. vector de Stock | Adobe Stock