Phosphorus-carbon bond forming reactions of iron tetracarbonyl-coordinated phosphenium ions - ScienceDirect

SOLVED: Which bond is weakest? Group of answer choices Carbon-Oxygen double bond Carbon-Phosphorus single bond Carbon-Nitrogen single bond Carbon-Oxygen triple bond Carbon-Nitrogen double bond

Phosphorus–Carbon Bond Forming Reactions of Diphenylphosphenium and Diphenylphosphine Triflate Complexes of Tungsten | Organometallics

The most common H-bond couplings in biomolecules. Hydrogen, nitrogen,... | Download Scientific Diagram

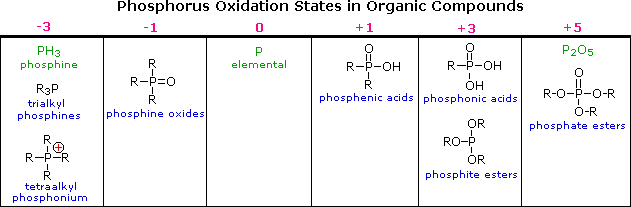
Recent advances in phosphorus–carbon bond formation: synthesis of H-phosphinic acid derivatives from hypophosphorous compounds - ScienceDirect

Isolation and Characterization of the Carbon–Phosphorus Bond-forming Enzyme Phosphoenolpyruvate Mutase from the MolluskMytilus edulis - ScienceDirect

Phosphorus–Carbon Bond Forming Reactions of Diphenylphosphenium and Diphenylphosphine Triflate Complexes of Tungsten | Organometallics

Pathways of carbon-phosphorus bond cleavage. (A) The C-P lyase pathway... | Download Scientific Diagram

Recent advances in phosphorus–carbon bond formation: synthesis of H-phosphinic acid derivatives from hypophosphorous compounds - ScienceDirect

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science
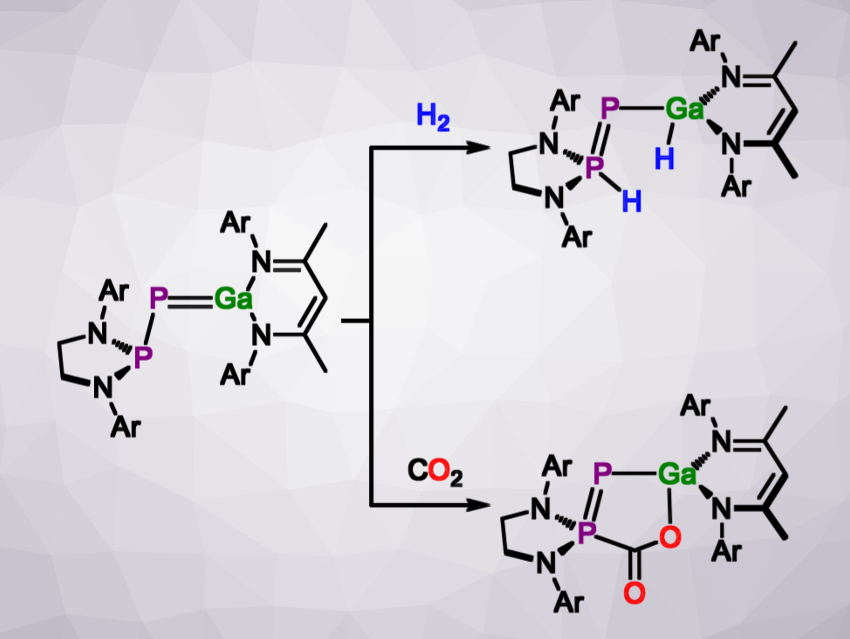
Recent advances in the carbon–phosphorus (C–P) bond formation from unsaturated compounds by s- and p-block metals - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB01019K

Phosphorus–Carbon Bond Forming Reactions of Diphenylphosphenium and Diphenylphosphine Triflate Complexes of Tungsten | Organometallics
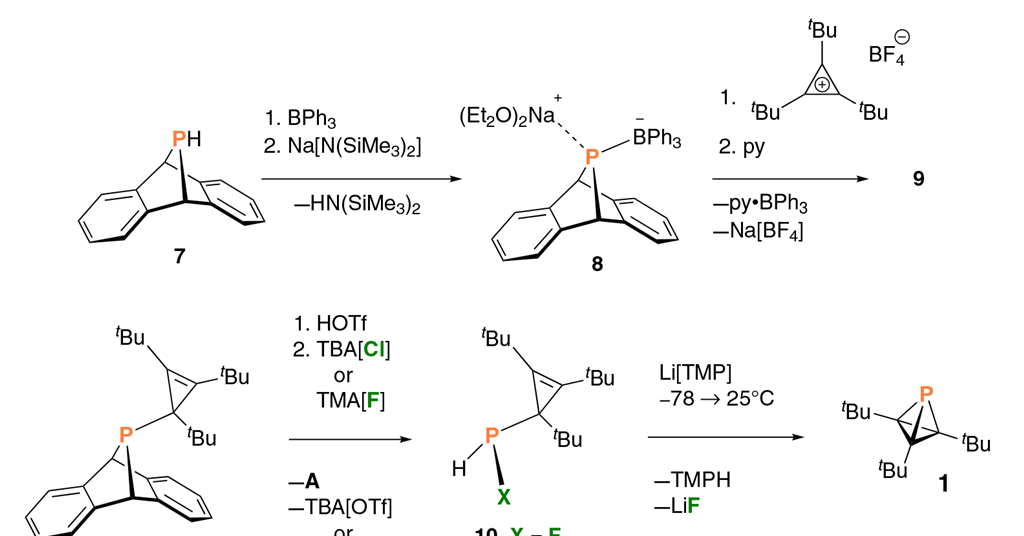
Carbon tetrahedron squeezes phosphorus into smallest possible bond angle | Research | Chemistry World

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science
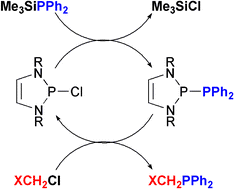
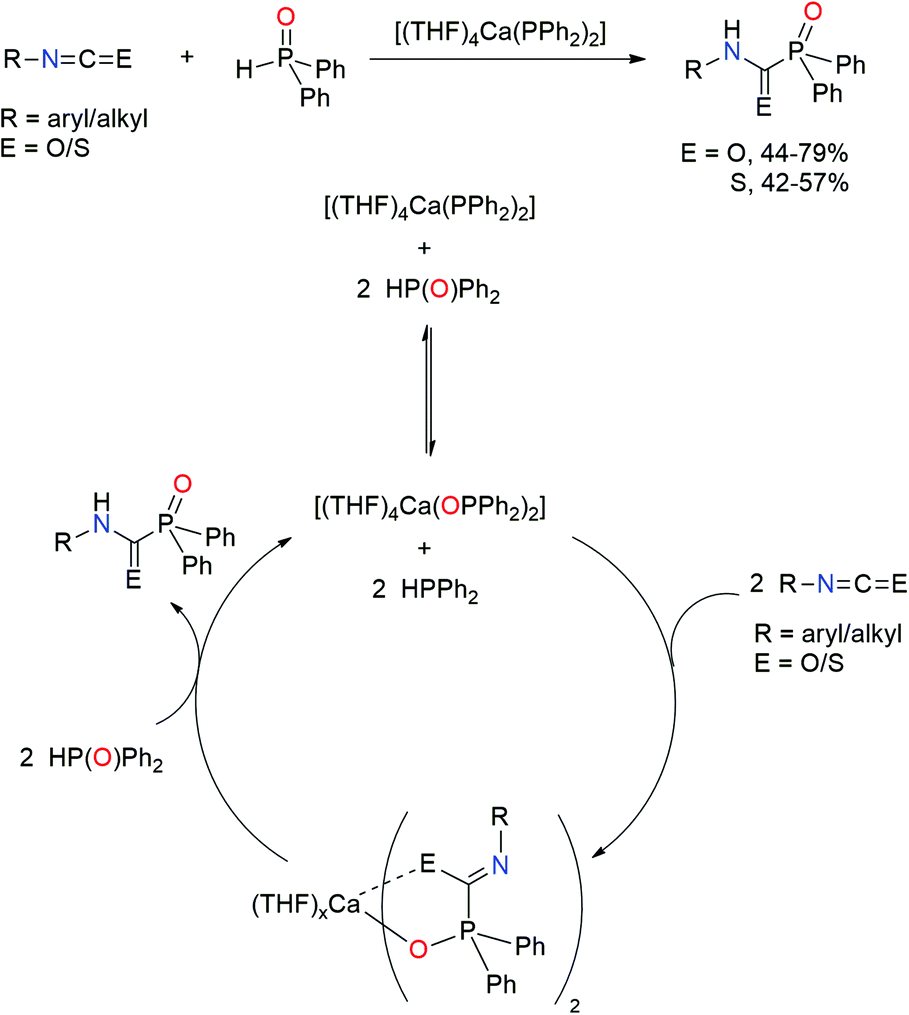
Phosphorus–carbon bond formation catalysed by electrophilic N-heterocyclic phosphines - Chemical Communications (RSC Publishing)
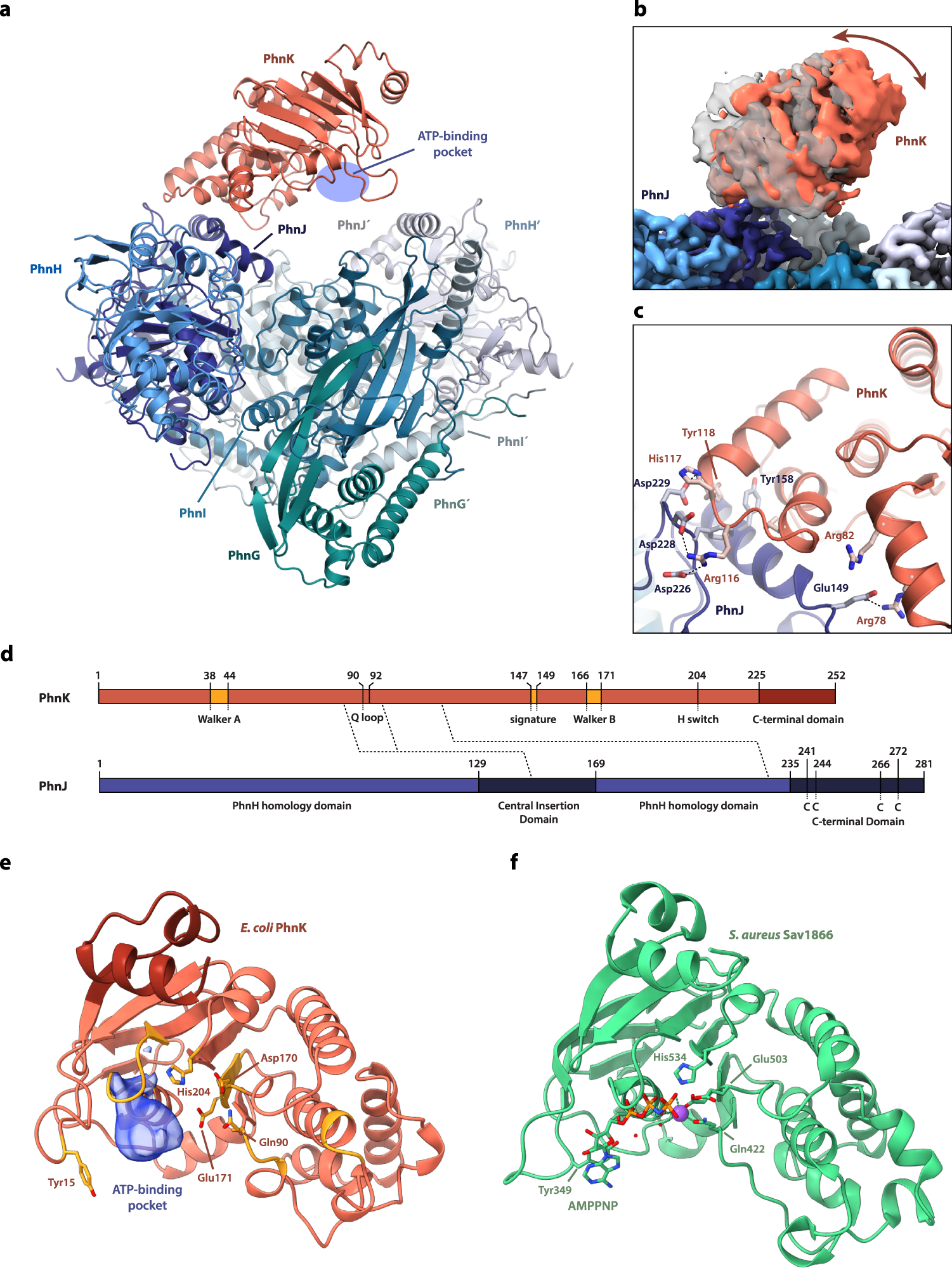
Structural remodelling of the carbon–phosphorus lyase machinery by a dual ABC ATPase | Nature Communications
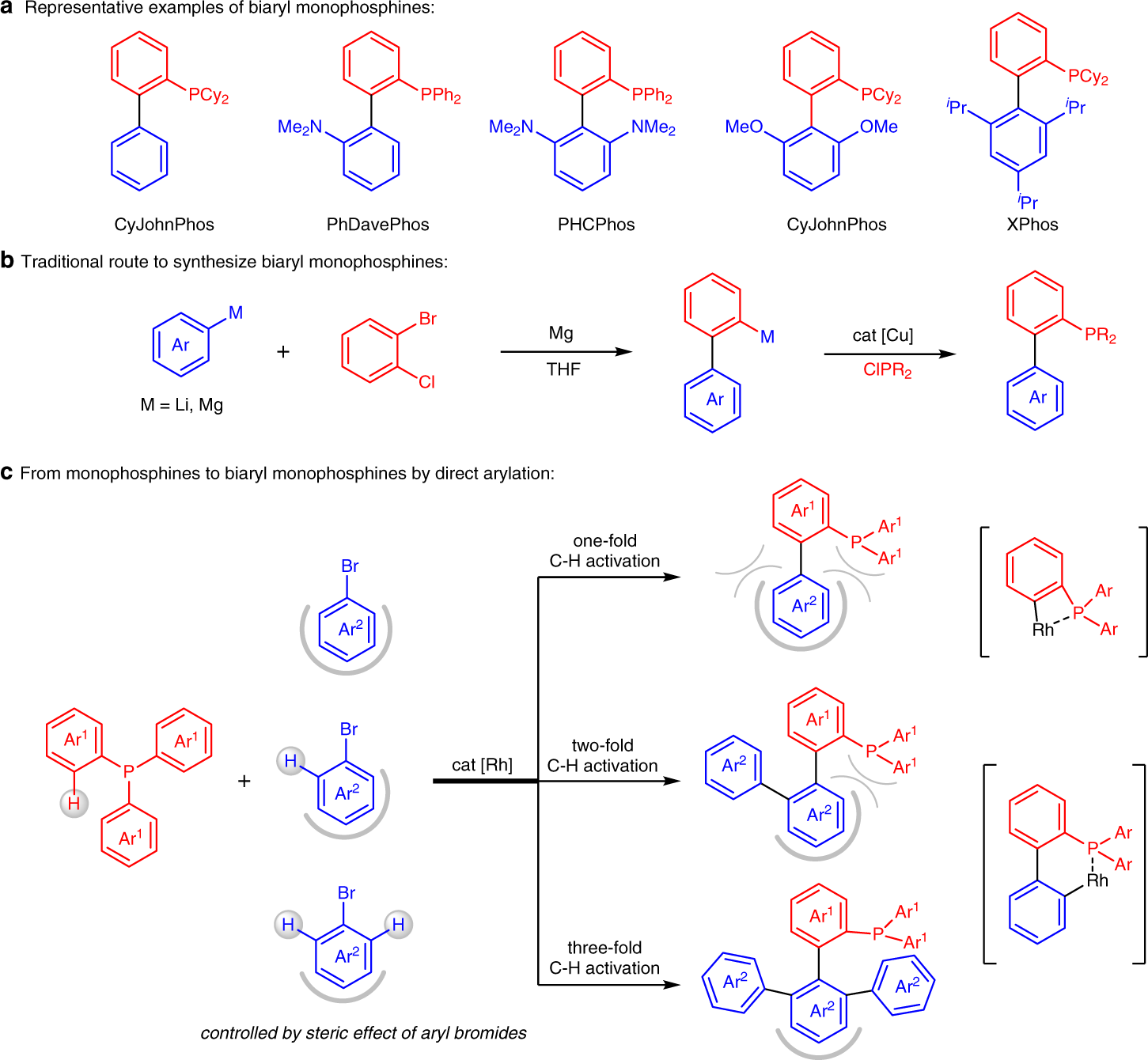
Rhodium-catalyzed selective direct arylation of phosphines with aryl bromides | Nature Communications
![PDF] Formation of stable phosphorus-carbon bond for enhanced performance in black phosphorus nanoparticle-graphite composite battery anodes. | Semantic Scholar PDF] Formation of stable phosphorus-carbon bond for enhanced performance in black phosphorus nanoparticle-graphite composite battery anodes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9c67d91d1f0476ca9912ff52569bfcfb4dde9d67/2-Figure1-1.png)