In the formation of Carbon tetra chloride molecule, state the following.a) Electronic configuration of nearest inert gas attained.b) How many electrons are sahred/ transferred in bond formation?c) Which type of bonds these
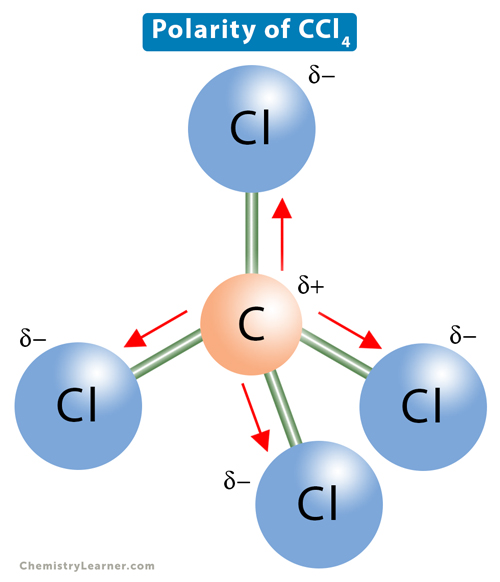
Carbon tetrachloride ($CC{{l}_{4}}$ ):(A) ionic substance(B) non-polar covalent substance(C) polar covalent substance (D) macromolecular substance(E) metallic substance
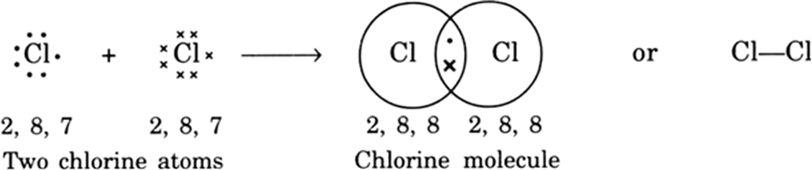
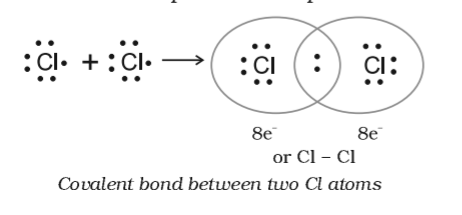
Explain the formation of covalent bonds in (i) chlorine molecule; (ii) carbon tetrachloride and (iii) ammonia. from Science Carbon and its Compounds Class 10 CBSE

b In the formation of carbon tetrachloride molecule, state the following:i Electronic configuration of nearest inert gas attained.ii How many electrons are shared/transferred in bond formationiii Which type of bonds these compounds

Answer the following:1) Write the formula and draw the electron dot structure of carbon tetrachloride.
Draw the structural formula of carbon tetrachloride and state the type of bond present in it - Sarthaks eConnect | Largest Online Education Community

In the formation of i Oxygen molecule ii Carbon tetrachloride molecule state the following a Electro...

Classify the following compounds as having covalent or ionic bonds: a) carbon tetrachloride b) rubidium oxide | Homework.Study.com

Explain the tetravalency of carbon with the help of electron dot structure - Science - Carbon and its Compounds - 10826407 | Meritnation.com

Explain the formation of Covalent compound.i) Chlorine Moleculesii) Carbon Tetra Chlorideiii) Ammonia - Brainly.in