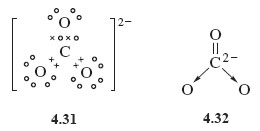
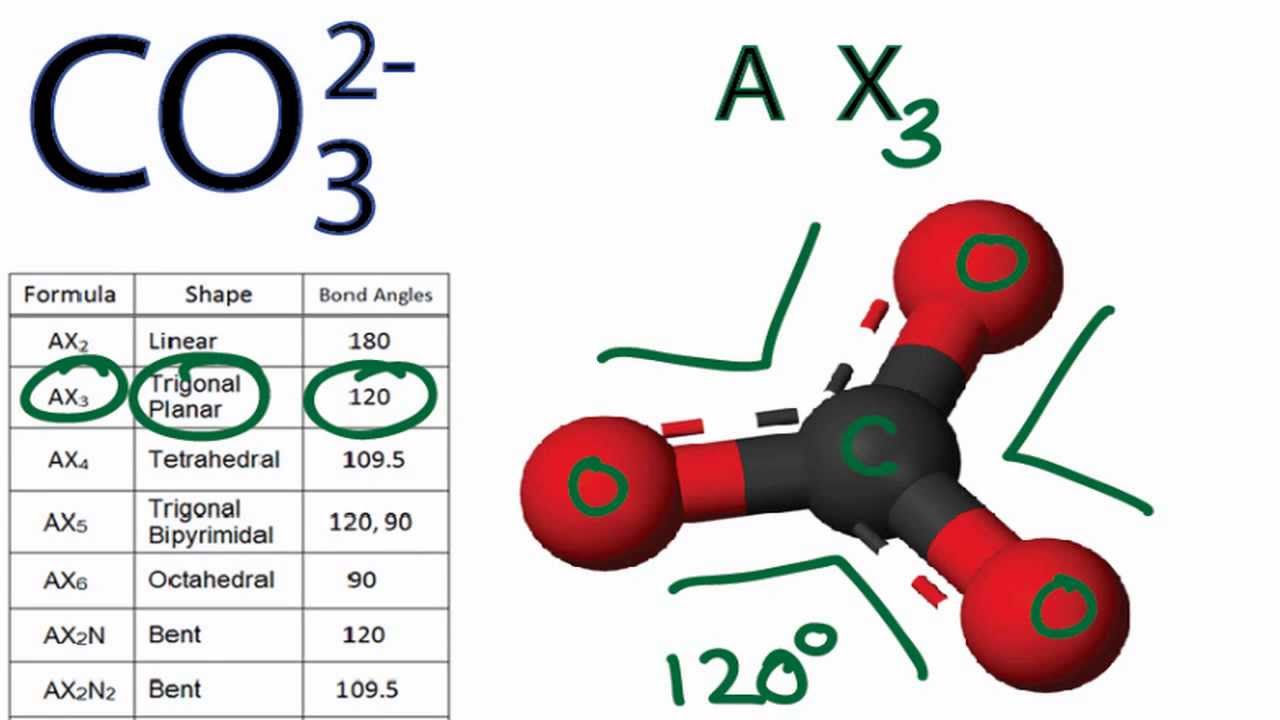
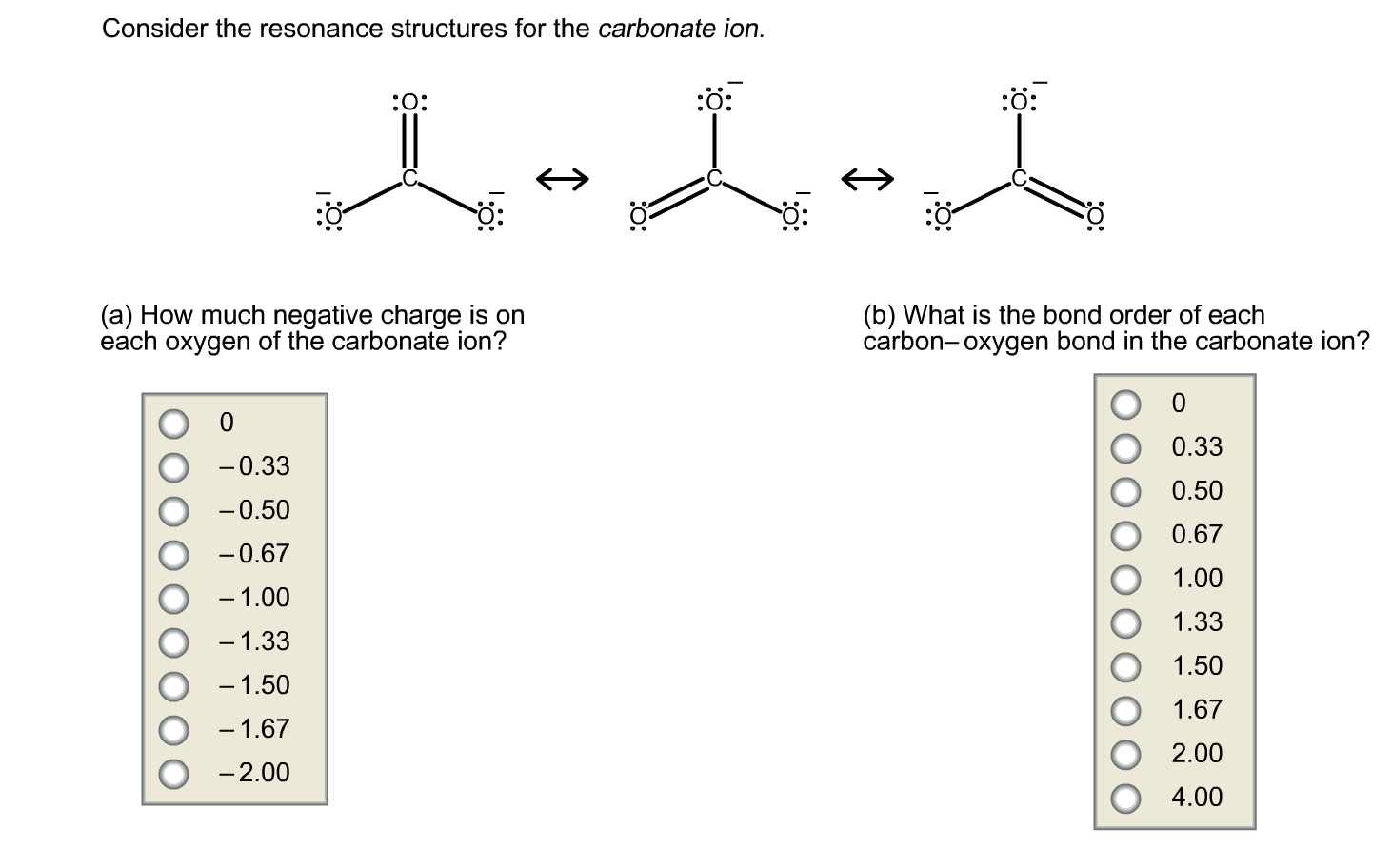
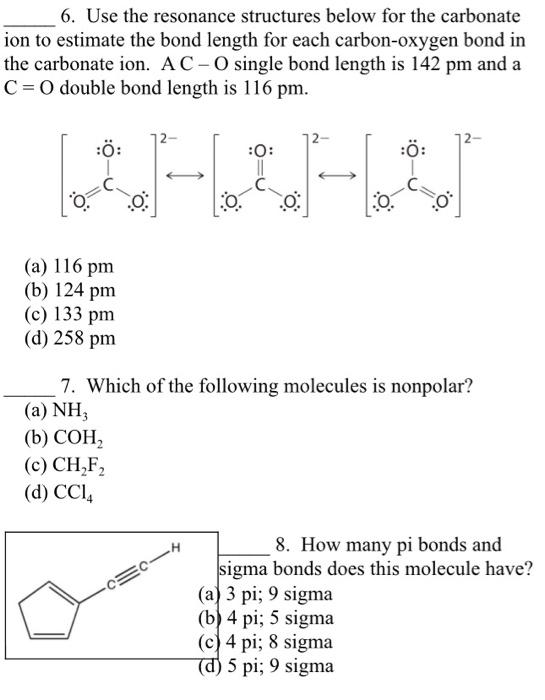
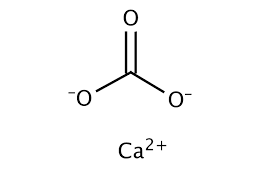
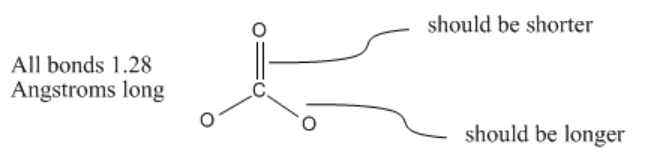
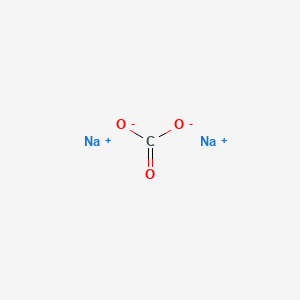
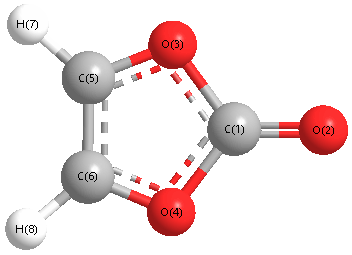
The completed Lewis structure of (CO3)2- contains a total of covalent bonds and lone pairs. Consider only a resonance structure in which all atoms have full octets. Note: If applicable, expand octets

Molecules | Free Full-Text | Chalcogen Bond Involving Zinc(II)/Cadmium(II) Carbonate and Its Enhancement by Spodium Bond

Chemistry - Chemical Bonding (21 of 35) Lewis Structures for Ions - Carbonate Ion - CO3(2-) - YouTube

Answer the bonding questions: A. In the carbonate ion, CO_3^2-, the total number of groups on the central atom is ...... (1, 2 ,3 ,4 , or some other number) B. In