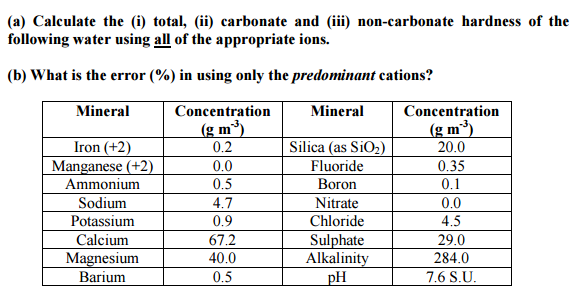
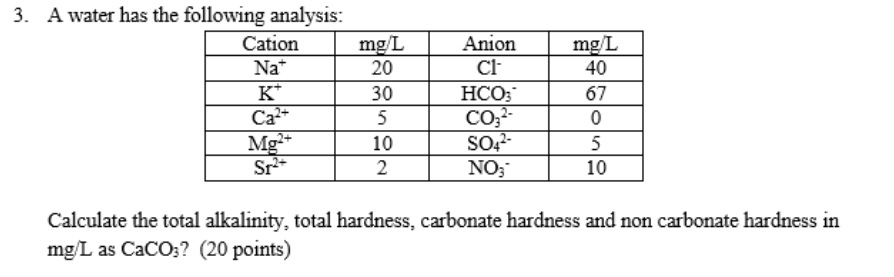
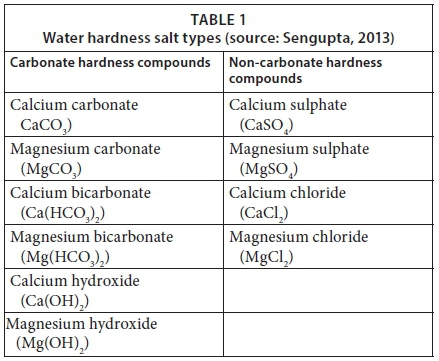
SOLVED: Problems Calculate total hardness, carbonate hardness, and non-carbonate hardness of the samples What is the degree ofhardness of the sample (sof, moderate; hard, very hard)? Why might we necd to know

3.pdf - Calculate total carbonate and non-carbonate hardness of 1L water sample which contains CaCl2 = 25.4 ppm MgSO4 = 23.6 ppm MgCl2 = 32 ppm | Course Hero
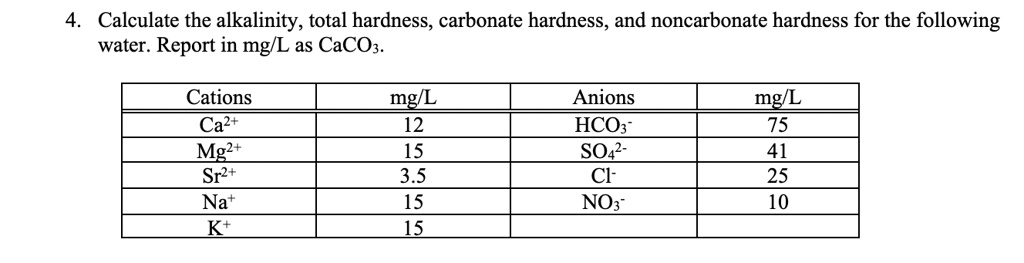
SOLVED: Calculate the alkalinity, total hardness, carbonate hardness, and noncarbonate hardness for the following water. Report in mg/L as CaCO3. Cations Ca2+ Mg2t Sr2+ Nat Kt mg/L 12 15 35 15 15
![SOLVED: Tuesday Aovemner [15] For the water analysis in the previous problem: Calculate the carbonate and non-carbonate hardness Calculate the lime dosage required for softening by selective calcium removal. Use 30 5 SOLVED: Tuesday Aovemner [15] For the water analysis in the previous problem: Calculate the carbonate and non-carbonate hardness Calculate the lime dosage required for softening by selective calcium removal. Use 30 5](https://cdn.numerade.com/ask_images/1fe181ed7edc46e1a636b320927fdd14.jpg)
SOLVED: Tuesday Aovemner [15] For the water analysis in the previous problem: Calculate the carbonate and non-carbonate hardness Calculate the lime dosage required for softening by selective calcium removal. Use 30 5