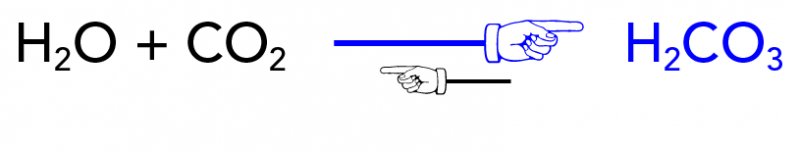
How does carbonation stay in soda unlike air which just bubbles up to the surface and doesn't seem to stay in liquid? - Quora
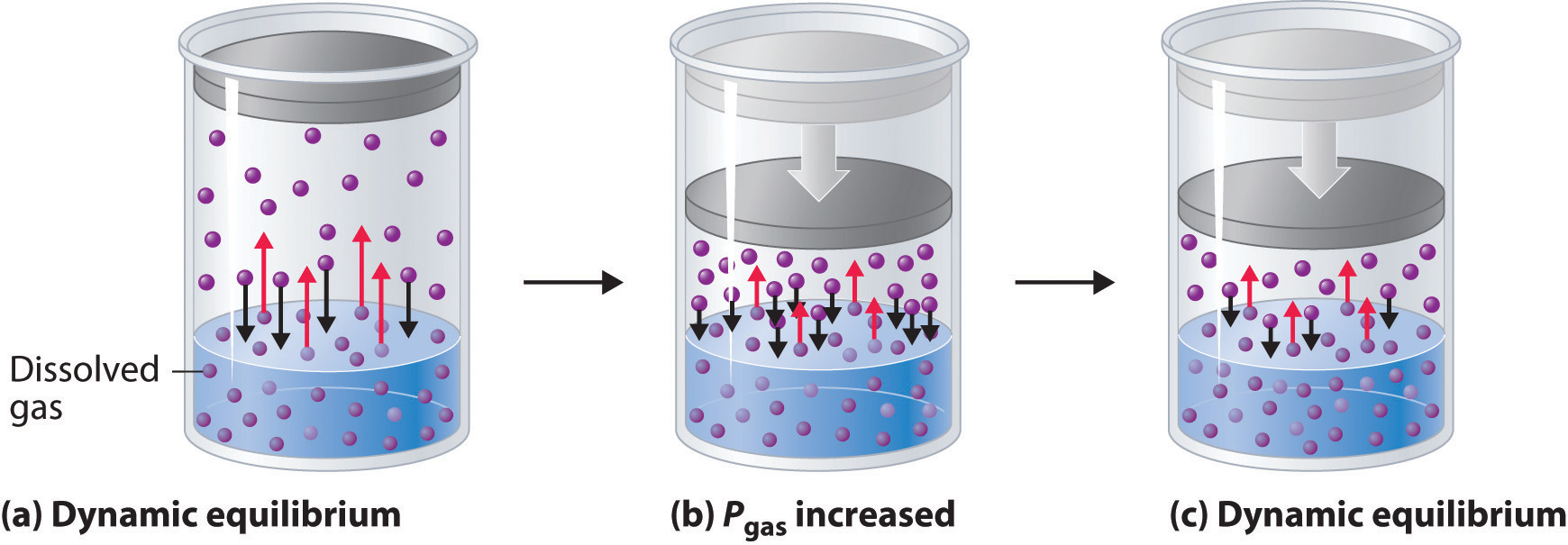
In a closed can of soda, dissolved carbonic acid is in equilibrium with CO2 and H2O. When the can is opened CO2 is produced until the pop goes flat. How do these