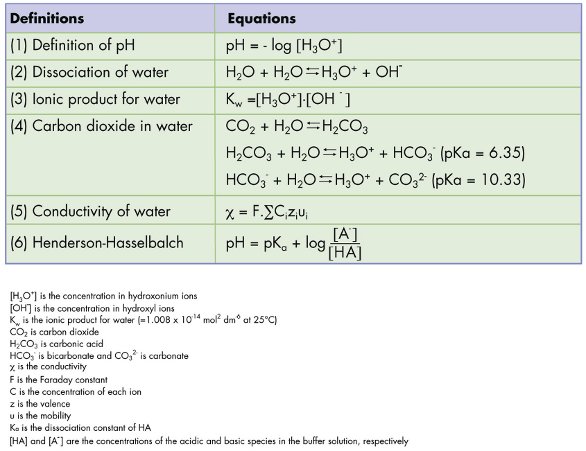
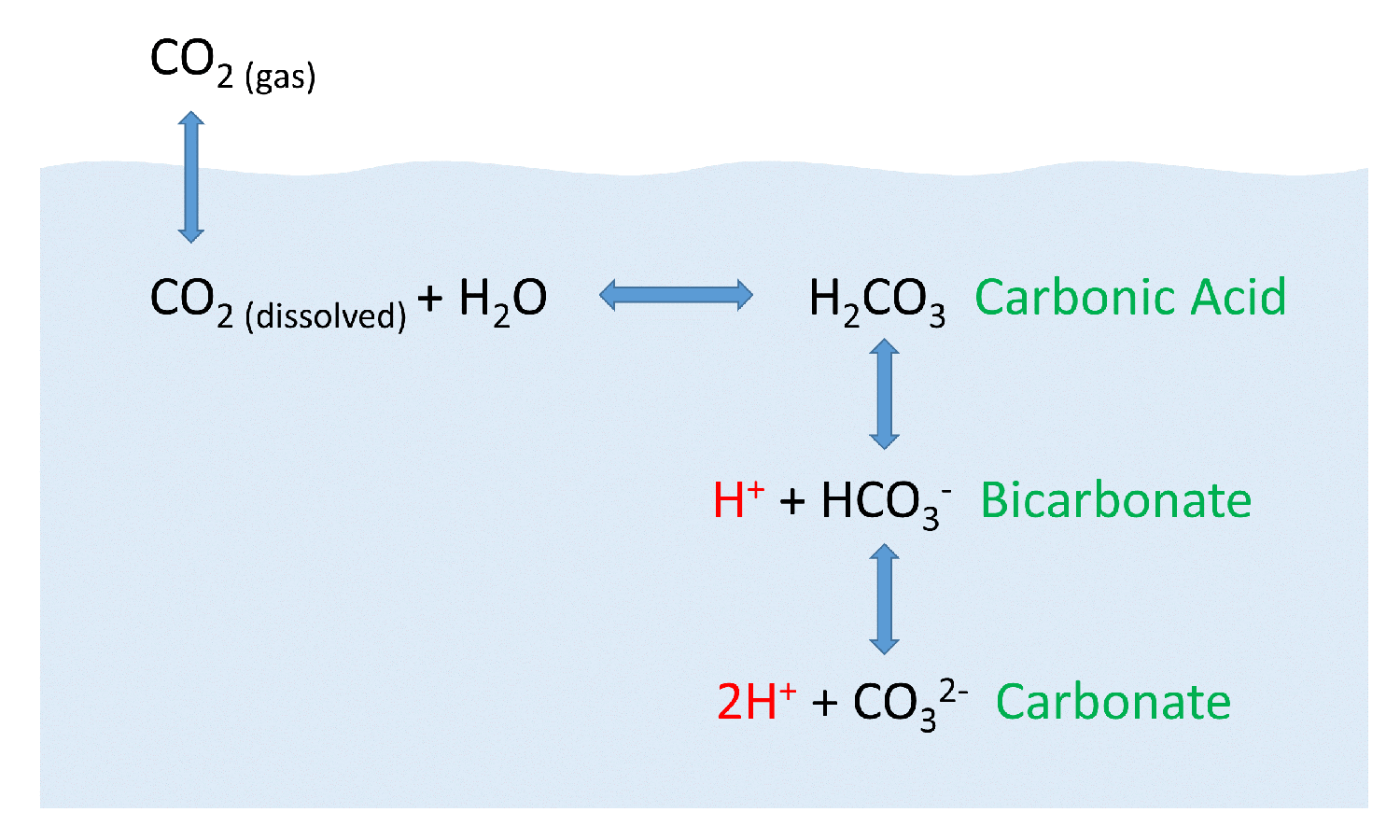
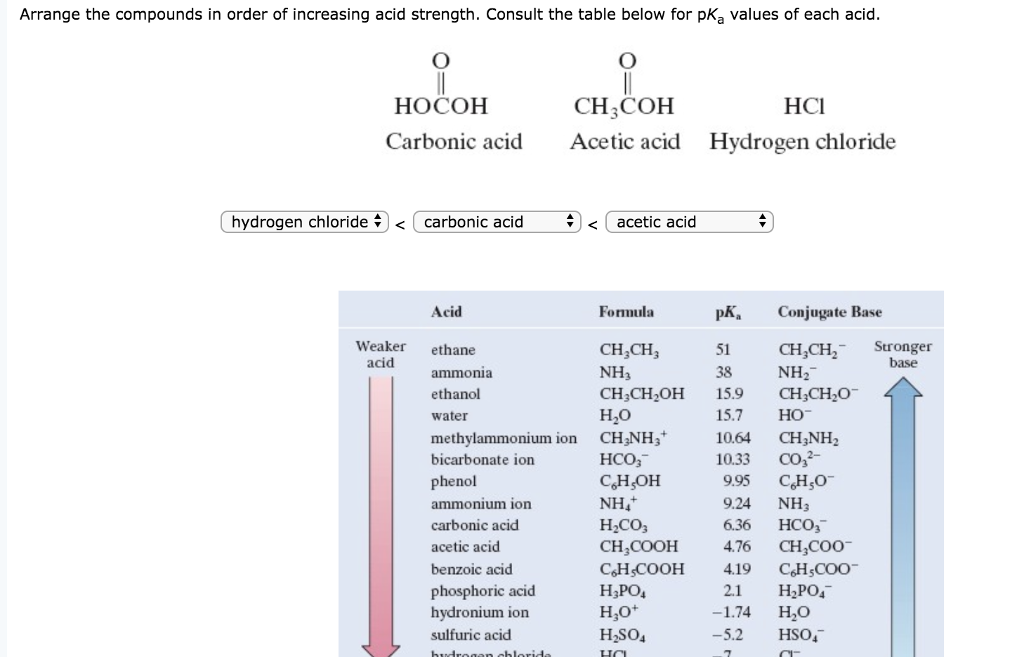
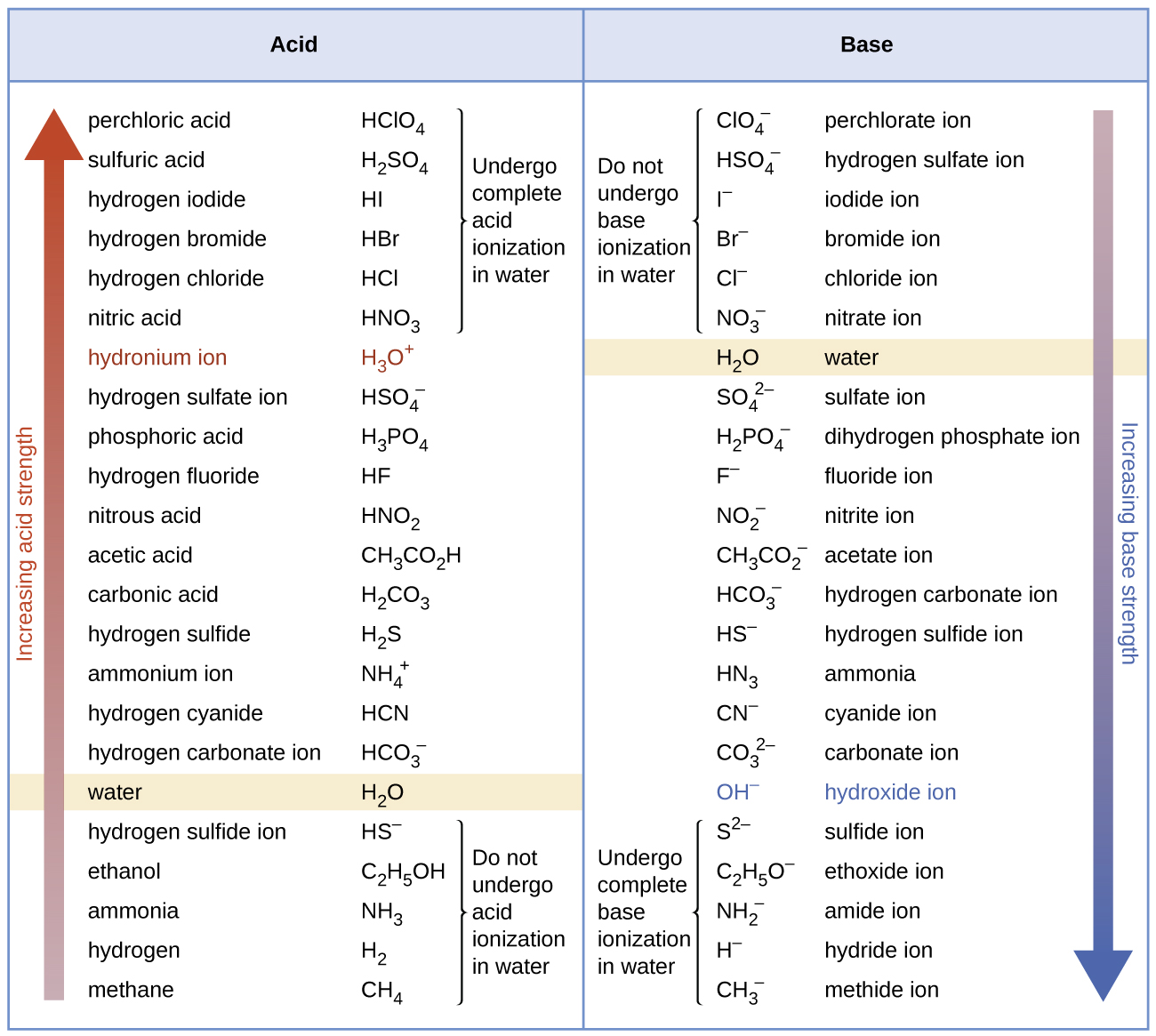
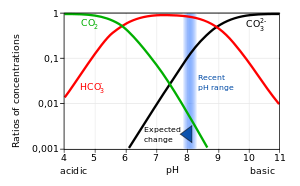
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora

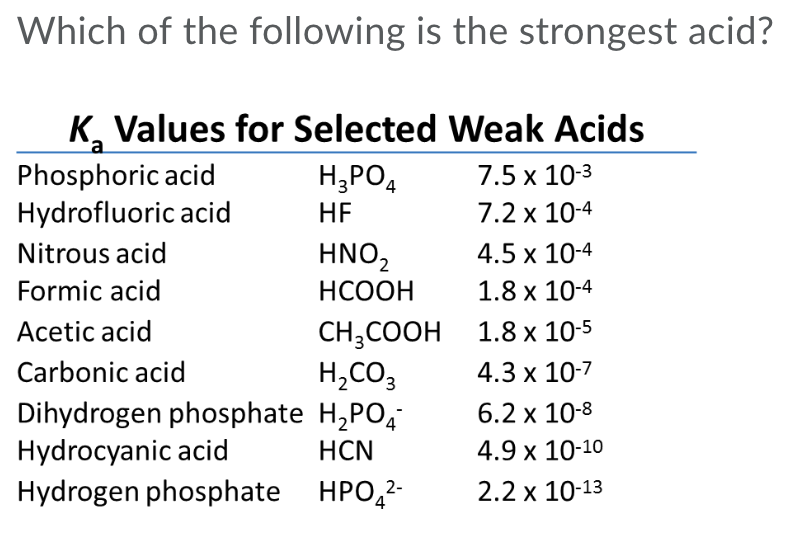
SOLVED: Part A - Acid Strength and Ka Carbonic acid (HzCOz) and formic acid (HCOzH) are two other weak acids Carbonic acid has Ka Of 4.3x 10" and formic acid has Ka