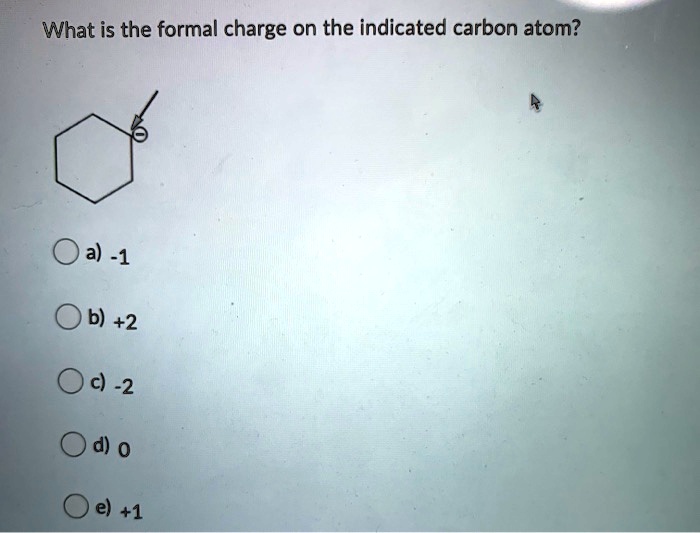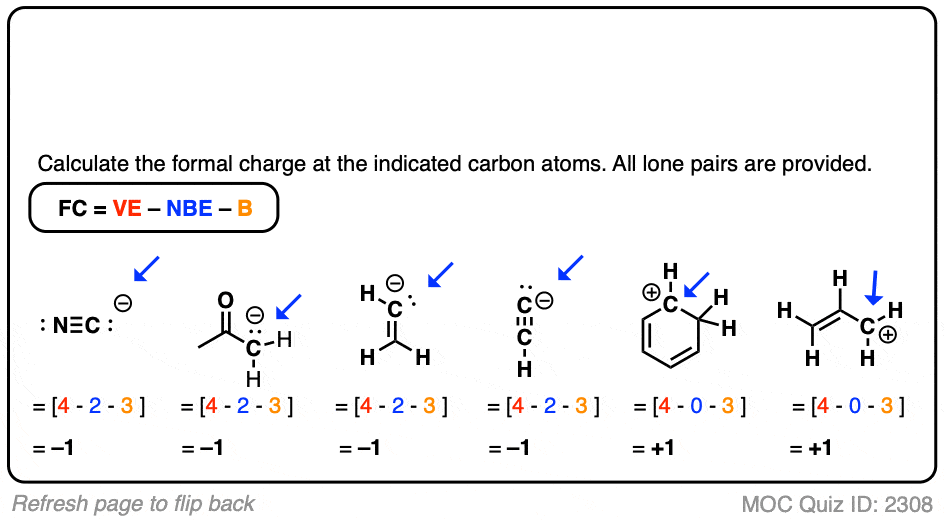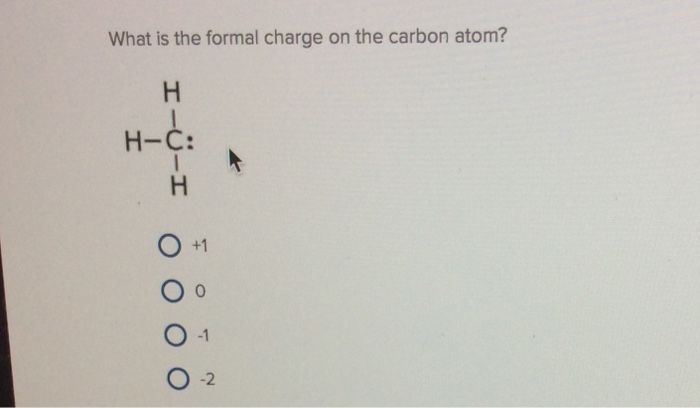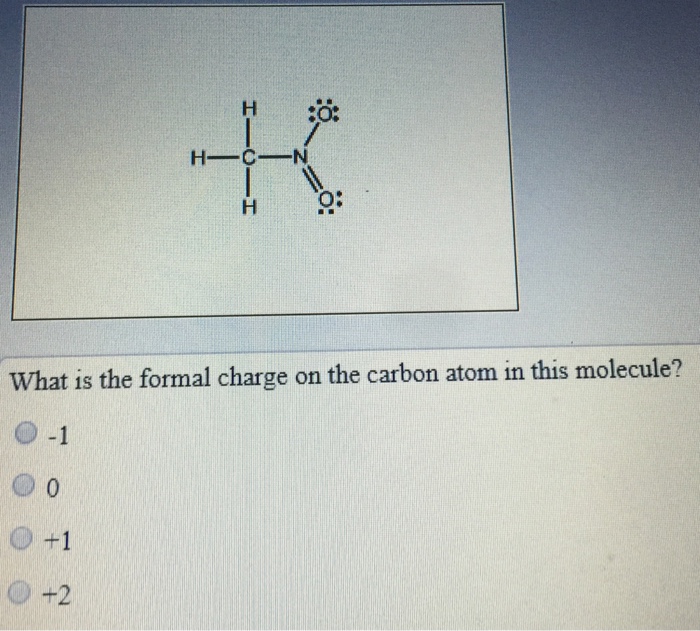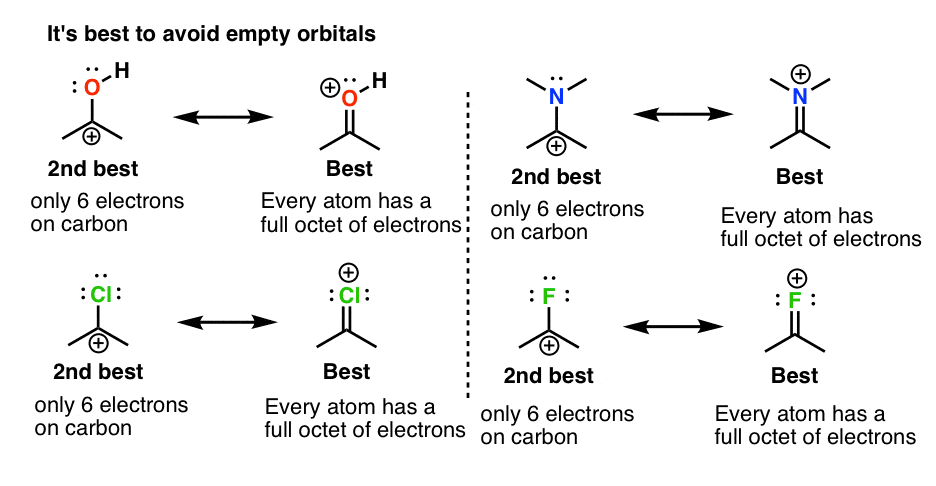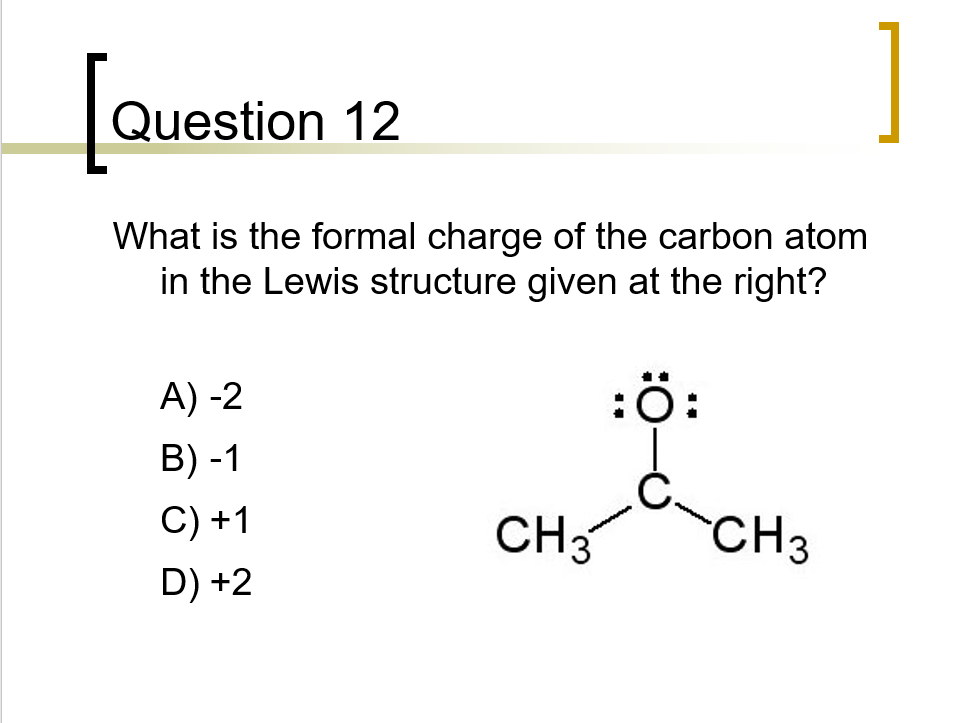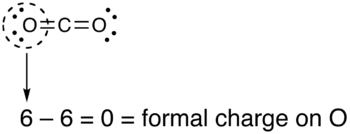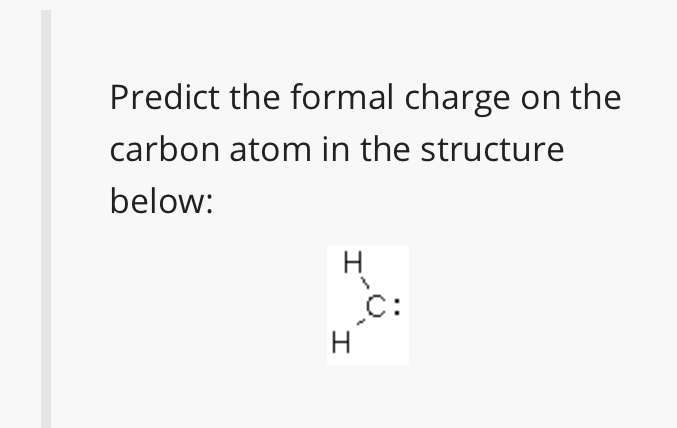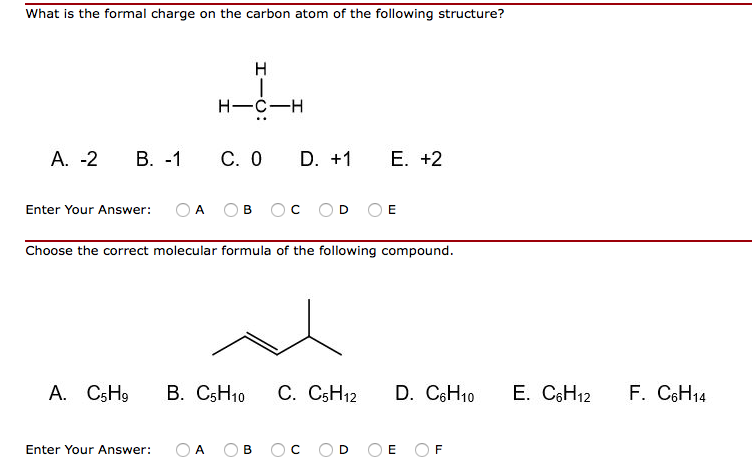
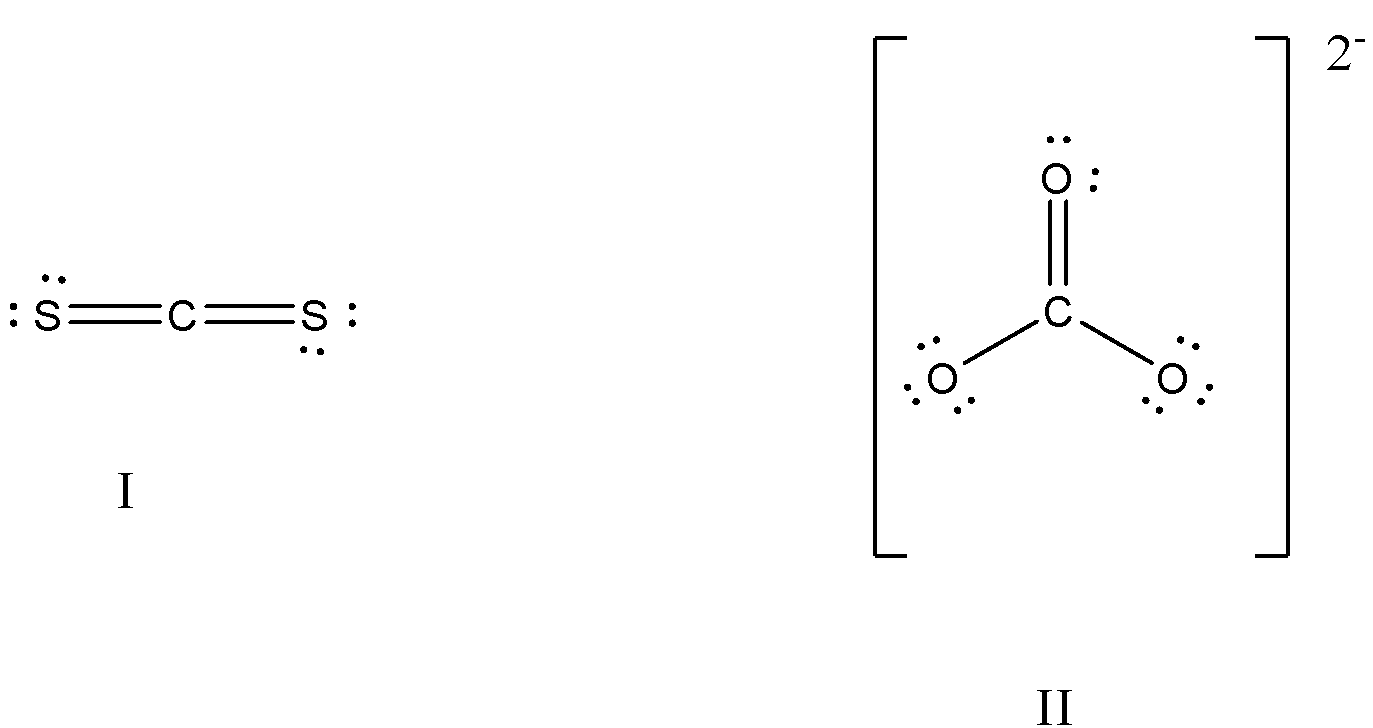
What is the formal charge on carbon atom in the following two structures are:\n \n \n \n \n \n \n \n \n \n A. 0,-2B. 0,0C. +2,-2D. +1,-1

Let R = {(1, 3), (4, 2), (2, 4), (2, 3), (3, 1)} be a relation on the set A = {1, 2, 3, 4} . The relation R is
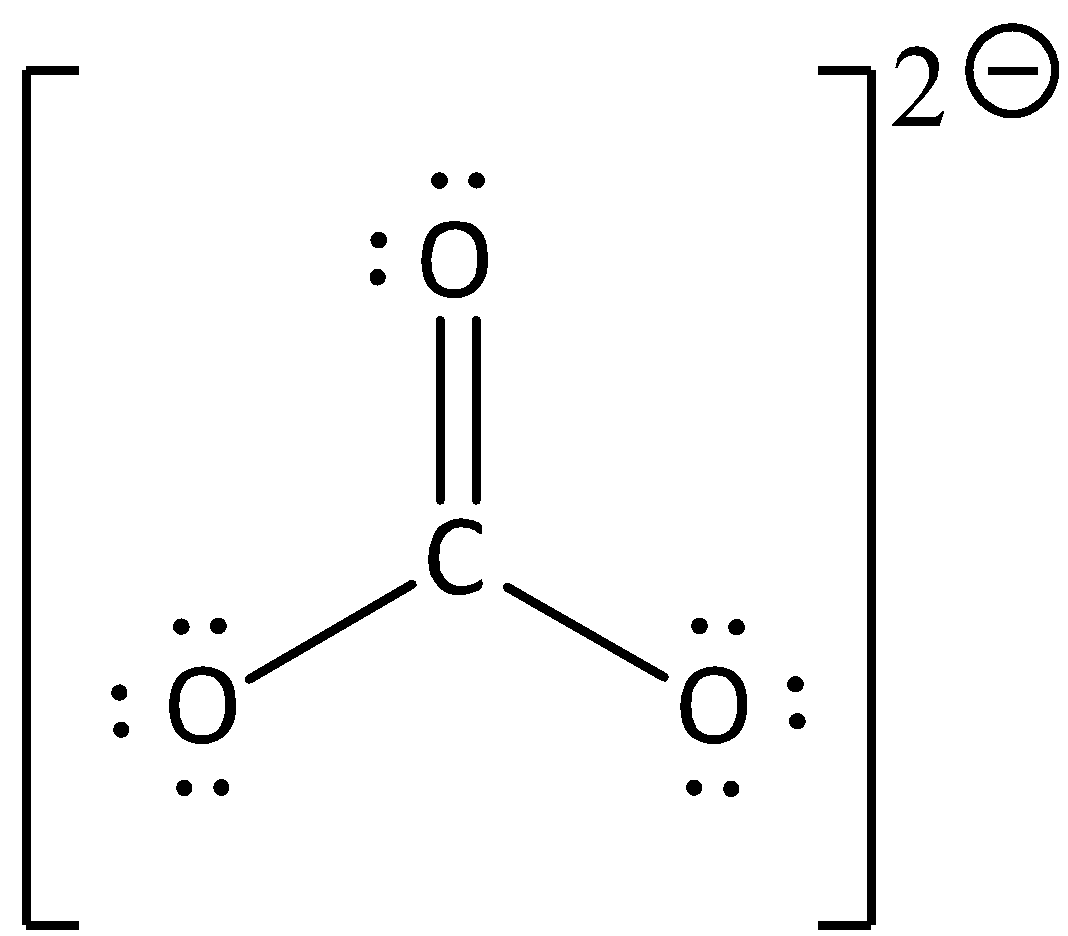
What is the formal charge on the carbon atom in the following two structures?\n \n \n \n \n (a)- 0, -2(b)- 0, 0(c)- +2, -2(d)- +1, -1

Relationship between natural charge of the carbon atom bonded to R 4... | Download Scientific Diagram
![Consider these struc [{Image src='img13134411427011773846060.jpg' alt='' caption=''}]tures. For which structure(s) does the electron-bearing carbon atom have a formal charge of zero? methylene only et | Homework.Study.com Consider these struc [{Image src='img13134411427011773846060.jpg' alt='' caption=''}]tures. For which structure(s) does the electron-bearing carbon atom have a formal charge of zero? methylene only et | Homework.Study.com](https://homework.study.com/cimages/multimages/16/img13134411427011773846060.jpg)
Consider these struc [{Image src='img13134411427011773846060.jpg' alt='' caption=''}]tures. For which structure(s) does the electron-bearing carbon atom have a formal charge of zero? methylene only et | Homework.Study.com