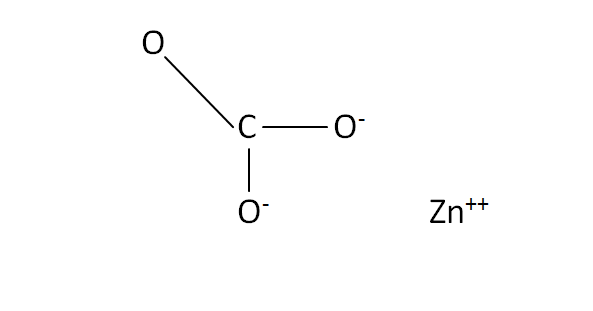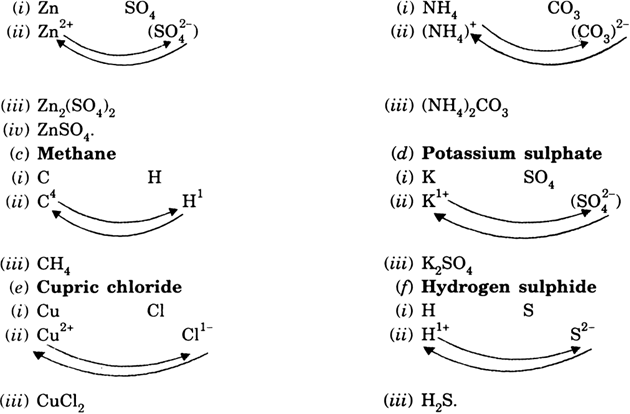
Write 'step-wise' the formulae for the following:(a) Zinc Sulphate (b) Ammonium carbonate(c) Methane (d) Potassium sulphide(e) Cupric chloride (f) Hydrogen sulphate. from Science Atoms And Molecules Class 9 CBSE

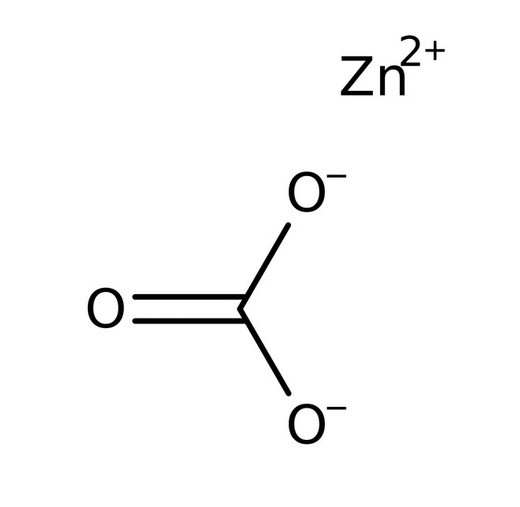
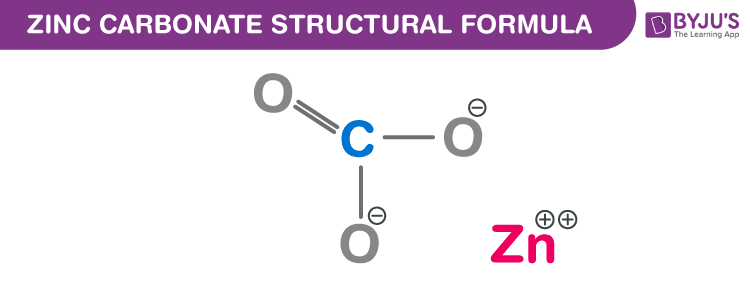
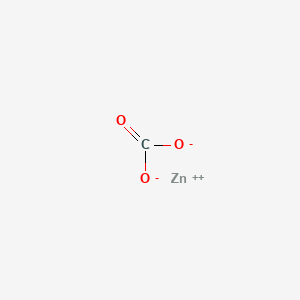
Write Molecular formula of Zinc Carbonate|Chemical formula of Zinc Carbonate |Zinc Carbonate - YouTube

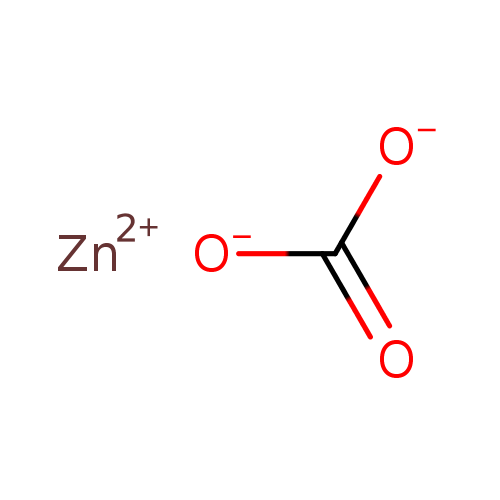
Question Video: Identifying the Correct Chemical Equation for the Decomposition of Zinc Carbonate | Nagwa

Write balanced chemical equations for the following word equation:Zinc sulphide + Oxygen → Zinc oxide + Sulphur dioxide

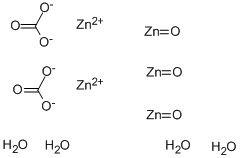
Write chemical reaction for each- (1) Zinc carbonate is calcinated. (2) cinnabar is heated in the air. (3) - Brainly.in