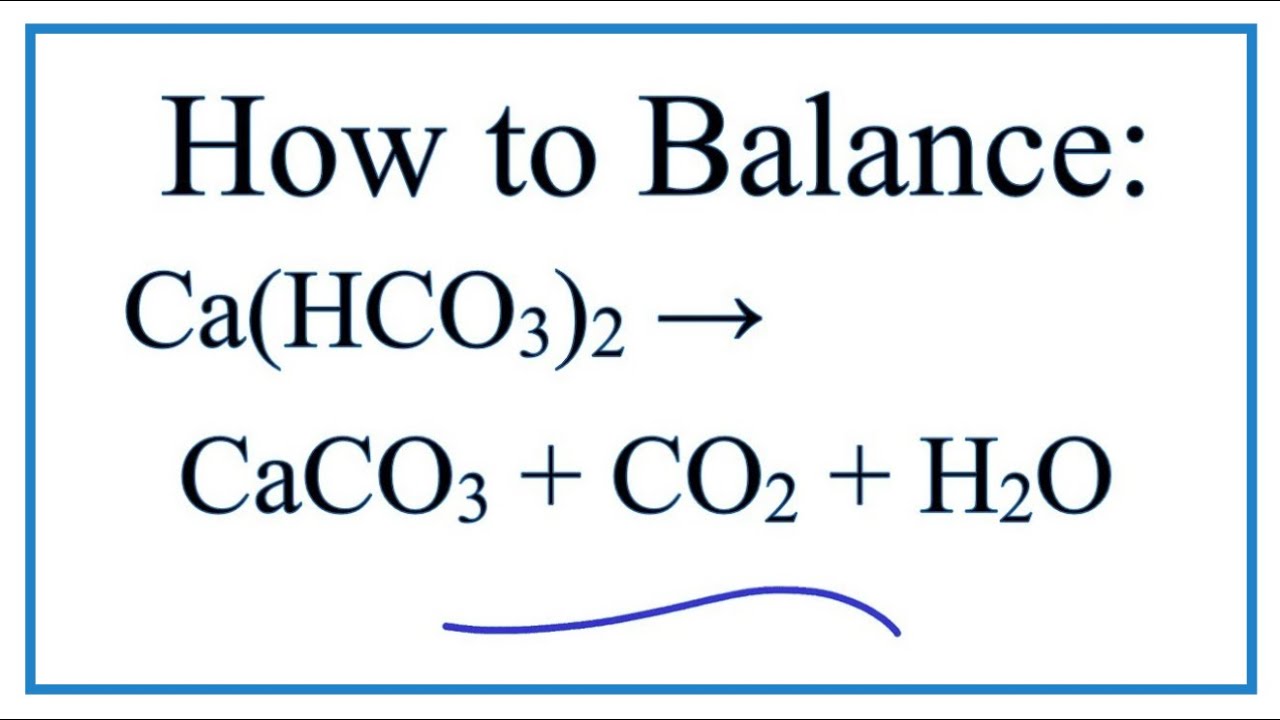
How to Balance Ca(HCO3)2 = CaCO3 + CO2 + H2O (Decomposition of Calcium hydrogen carbonate) - YouTube

Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram
![SOLVED: What is the expression for the equilibrium constant? CaO(s) + CO2(g) ⇌ CaCO3(s) [CaO][CO2] / [CaCO3] b) Keq = [CaCO3] / [CaO][CO2] c) Keq = [ CO2] [CO2] Keq = [CO2] Keq = [ SOLVED: What is the expression for the equilibrium constant? CaO(s) + CO2(g) ⇌ CaCO3(s) [CaO][CO2] / [CaCO3] b) Keq = [CaCO3] / [CaO][CO2] c) Keq = [ CO2] [CO2] Keq = [CO2] Keq = [](https://cdn.numerade.com/ask_images/928a15231b094c709901e0e2efd39d00.jpg)
SOLVED: What is the expression for the equilibrium constant? CaO(s) + CO2(g) ⇌ CaCO3(s) [CaO][CO2] / [CaCO3] b) Keq = [CaCO3] / [CaO][CO2] c) Keq = [ CO2] [CO2] Keq = [CO2] Keq = [
In the equilibrium CaCO3(s)Cao (s)+CO2(g) at 1073 K, the pressure of CO2 isfound to be 2.5 x 10 Pa. The equilibriumconstant for the reaction at 1073 K will be.(a) 0.25(c)25(b) 2.5(d) 250

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

Question Video: Determining the Expression for the Equilibrium Constant for the Decomposition of Calcium Carbonate | Nagwa

Reaction steps involved in the formation of the CaCO3 sol from calcium... | Download Scientific Diagram
![CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is](https://dwes9vv9u0550.cloudfront.net/images/10263170/3e36f846-ca64-4ea9-be05-3cab519e4454.jpg)
CaCO3→ CaO + CO2,Δ H = + 180KJ . If entropies of CaCO3,CaO and CO2 respectively are 93,39 and 213J/mol/K , then Δ S (total) [in J/K] at 300K is
If 220 grams of calcium oxide (CaO) reacts with 50L of carbon dioxide (CO2), what mass of calcium carbonate (CaCO3) is produced? - Quora
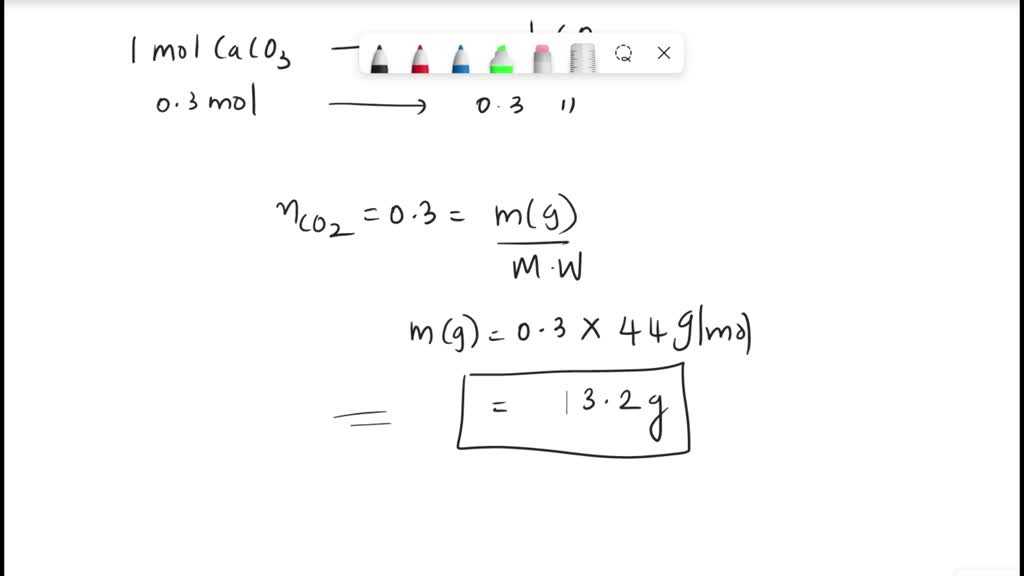
SOLVED: The compound CaCO3 can be decomposed to form CaO and CO2. This can be represented at the atomic level by a chemical equation: CaCO3 â†' CaO + CO2. According to the



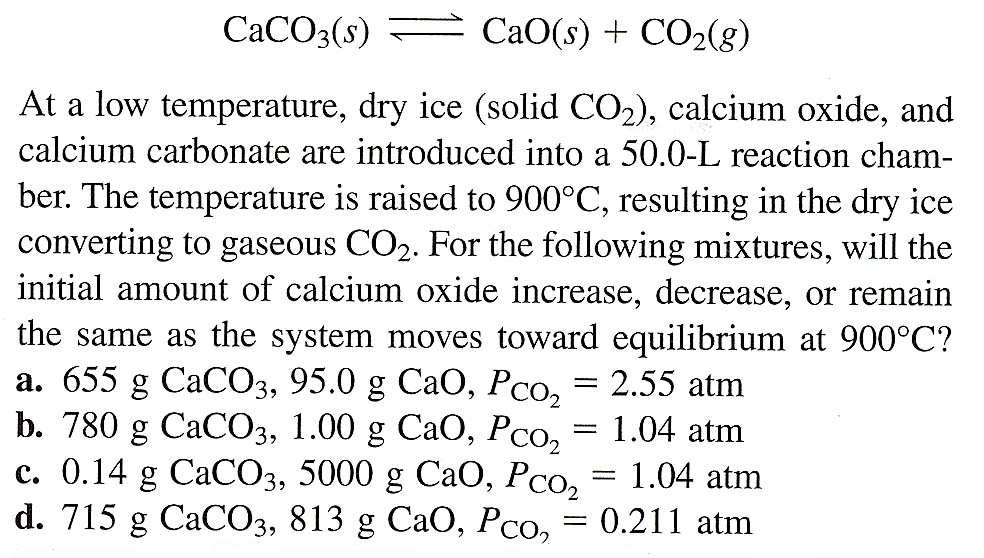







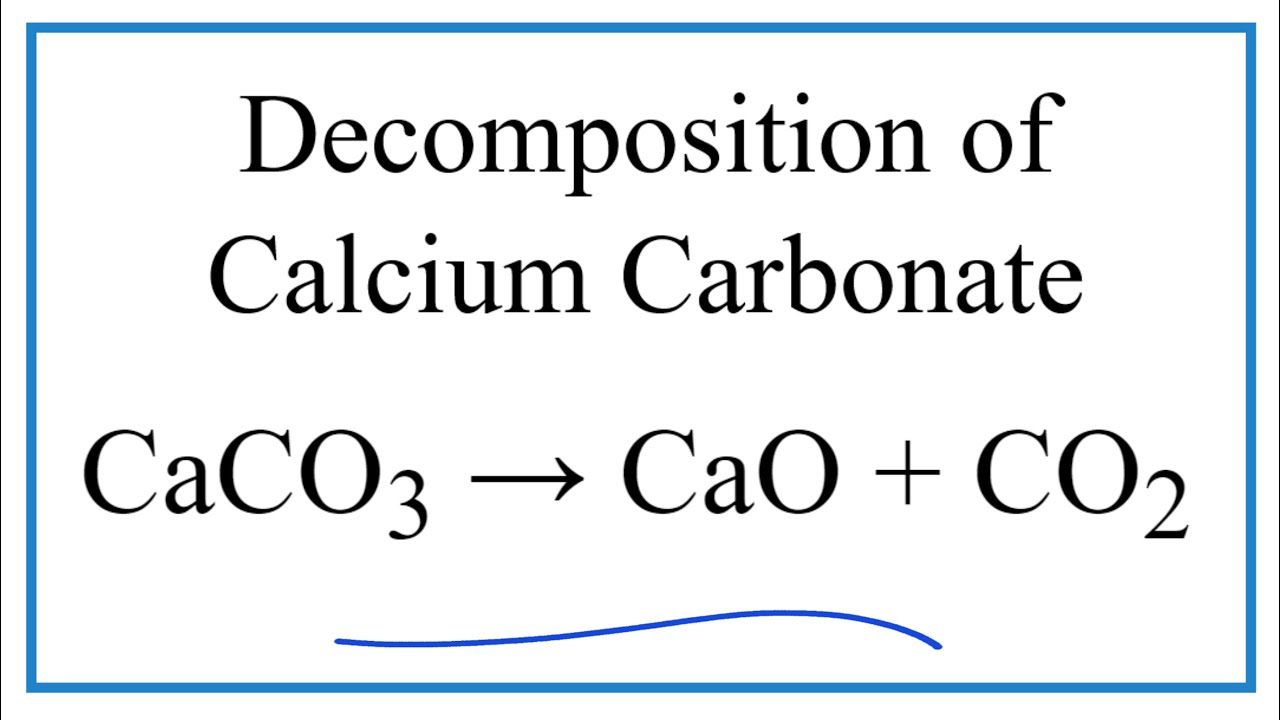


![ANSWERED] Given the following chemical equation CaO... - Physical Chemistry ANSWERED] Given the following chemical equation CaO... - Physical Chemistry](https://media.kunduz.com/media/sug-question-candidate/20220517131515311837-4392391.jpg)