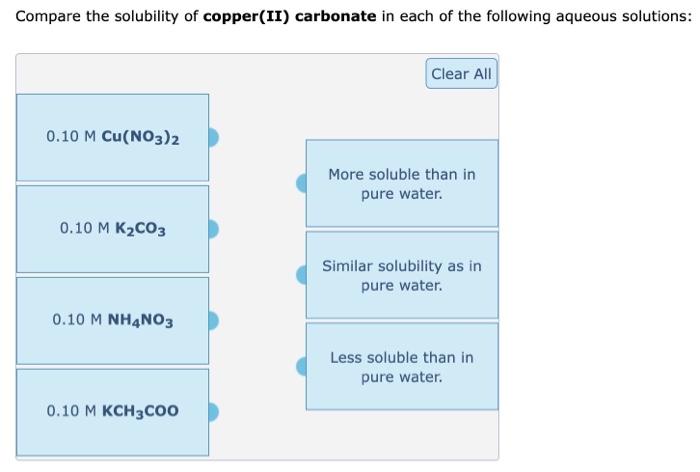
Aqueous Solutions. Soluble and Insoluble Soluble generally means that more than 1 g of solute will dissolve in 100 mL of water at room temperature. Insoluble. - ppt download

Copper (II) carbonate (CuCO3) powder in a crucible. This is a toxic green solid, which is soluble in acids but insoluble in water & alcohol Stock Photo - Alamy
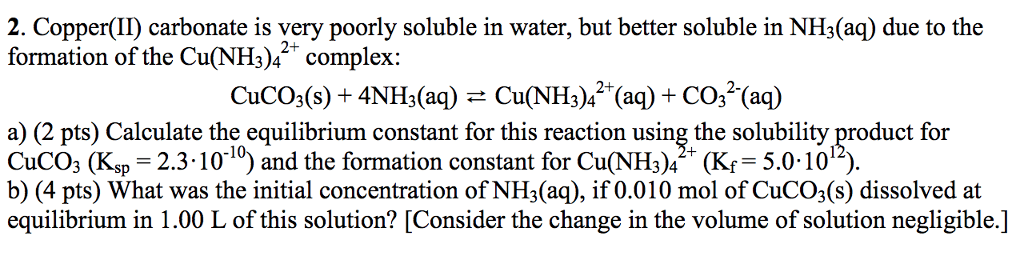
OneClass: 2. Copper(I) carbonate is very poorly soluble in water, but better soluble in NH3(aq) due t...
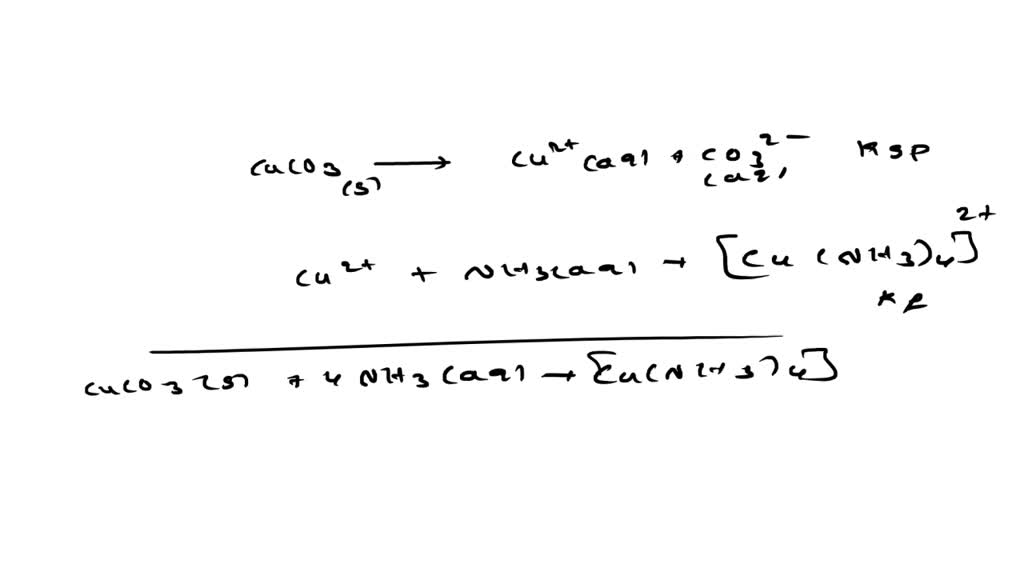
SOLVED: Copper carbonate is not very soluble in water, but it will dissolve in ammonia solutions because it forms a complex ion. Given the equilibrium constants below, what is the solubility of






![Solubility of Salts [Online Video] – O Level Secondary Chemistry Tuition Solubility of Salts [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2019/02/salt-solubility.jpg?w=1200)