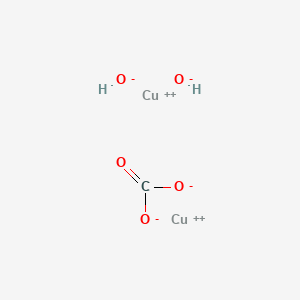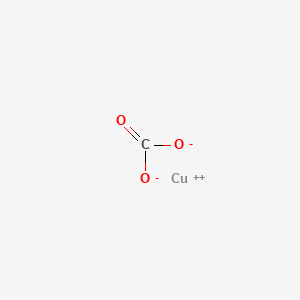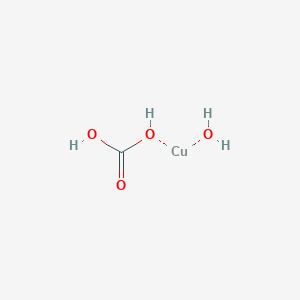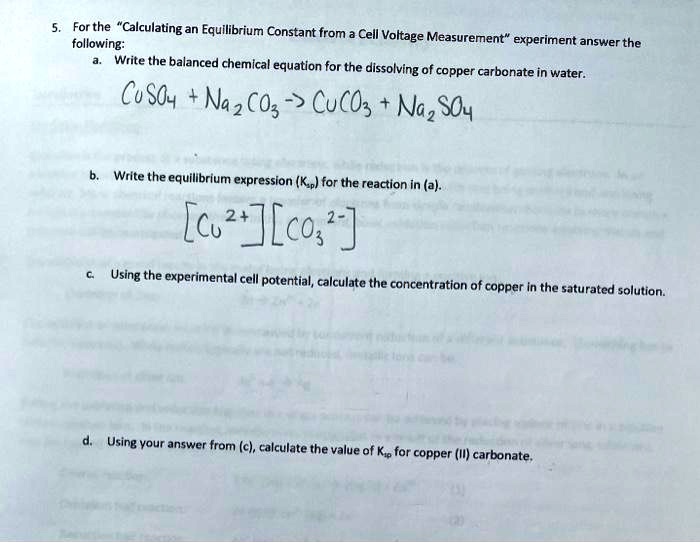
SOLVED: For the "Calculating an Equilibrium Constant from following: Cell Voltage Measurement" experiment answer the Write the balanced chemical equation for the dissolving of = copper carbonate in water: CoSOy NazC C0;

Carbon Dioxide from Thermal Decomposition (2.3.4) | Edexcel IGCSE Chemistry Revision Notes 2019 | Save My Exams

What is the Difference Between Copper Carbonate and Basic Copper Carbonate | Compare the Difference Between Similar Terms

Write a balanced equation for the preparation of the following salts.(i) Copper sulphate from Copper carbonate.(ii) Zinc carbonate from zinc sulphate.