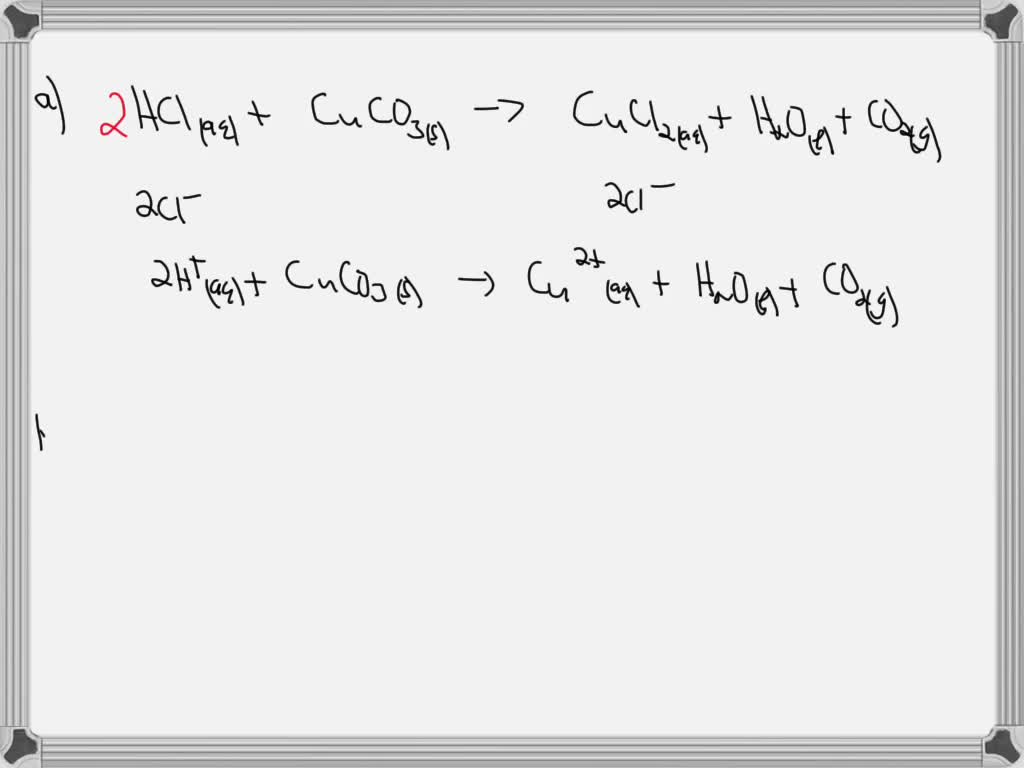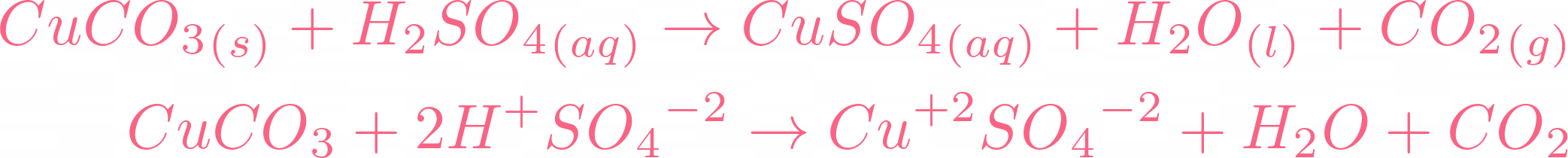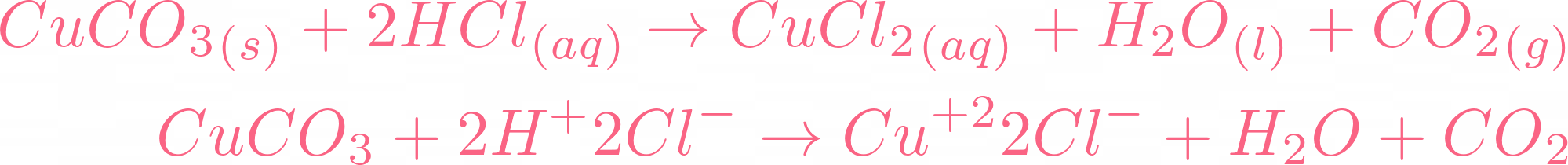Colour changes for simple indicators IndicatorColour in acidColour in alkali litmusredblue methyl orangeredyellow phenolphthaleincolourlessred. - ppt download
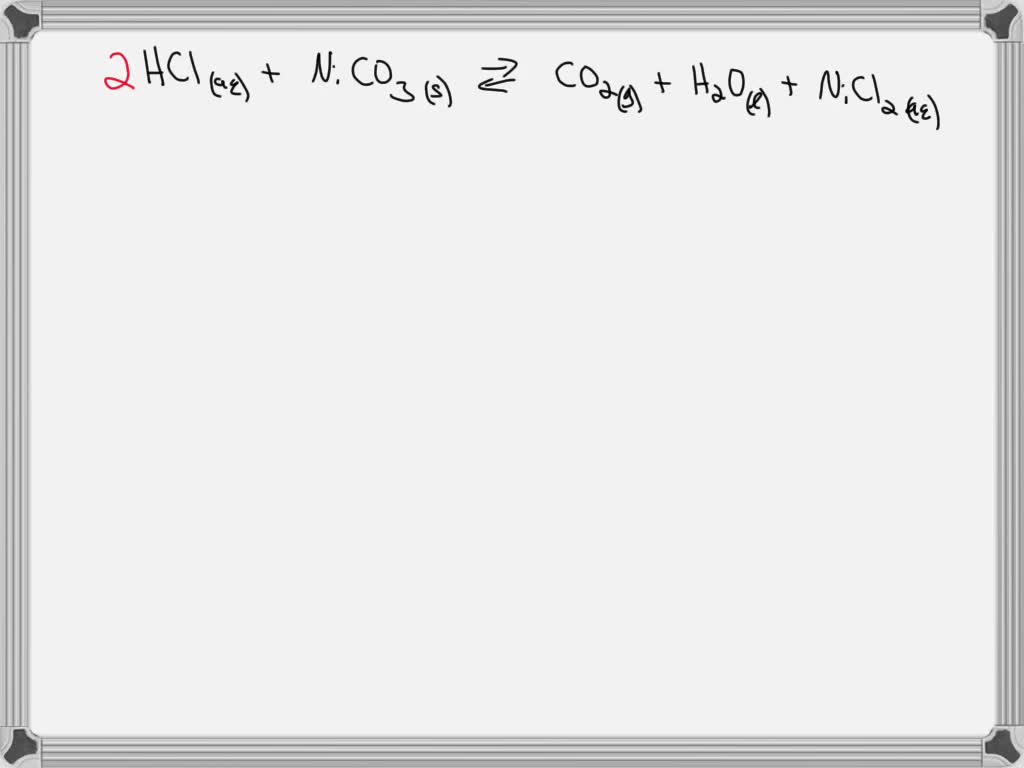
SOLVED: Write a net ionic equation for the reaction that occurs when solid copper(II) carbonate is combined with excess aqueous hydrobromic acid .

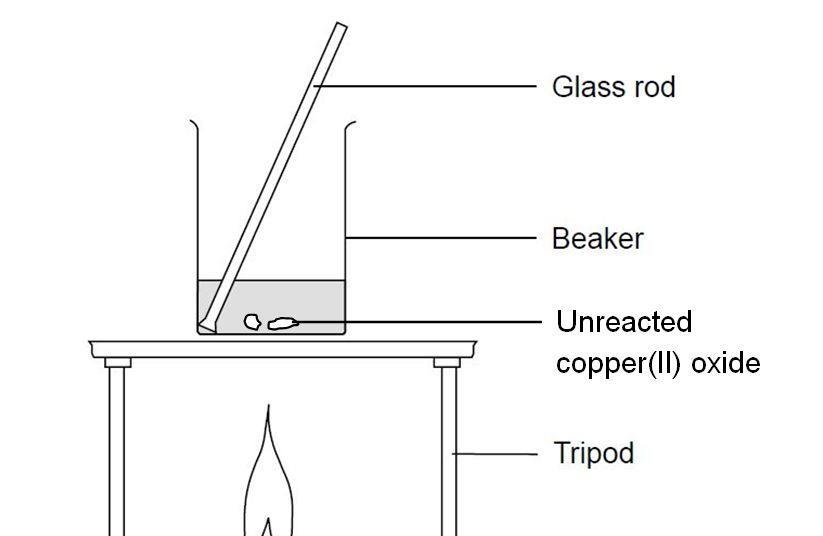
Copper Carbonate,Senior Chemistry - Extended Experimental In-Industry News-Nickel Acetate,Cobalt Sulfate-Fairsky Industrial Co., Limited

SOLVED: Copper carbonate (CuCO3) reacts with hydrochloric acid (HCl) according to this equation: CuCO3(s) + 2HCl(aq) → CuCl2(aq) + H2O(l) + CO2(g). Which statement correctly describes the substances in this reaction? A.

Acids and Alkalis Year 11. CONTENTS Acidity and alkalinity Indicators pH Acids General methods for making salts Making salts from metal oxides Making. - ppt download

How would you write the name of the following compounds ◦ Zn(OH) 2 ◦ NaOH ◦ HCl ◦ Mg(NO 3 ) 2 What does an acid do to red litmus? What does an acid to. - ppt download

SOLVED: Question 14 pts Solid copper (II) carbonate Is added t0 an aqueous solution of hydrochloric acid. Predict the products of this reaction: Write the molecular equation and net ionic equation for