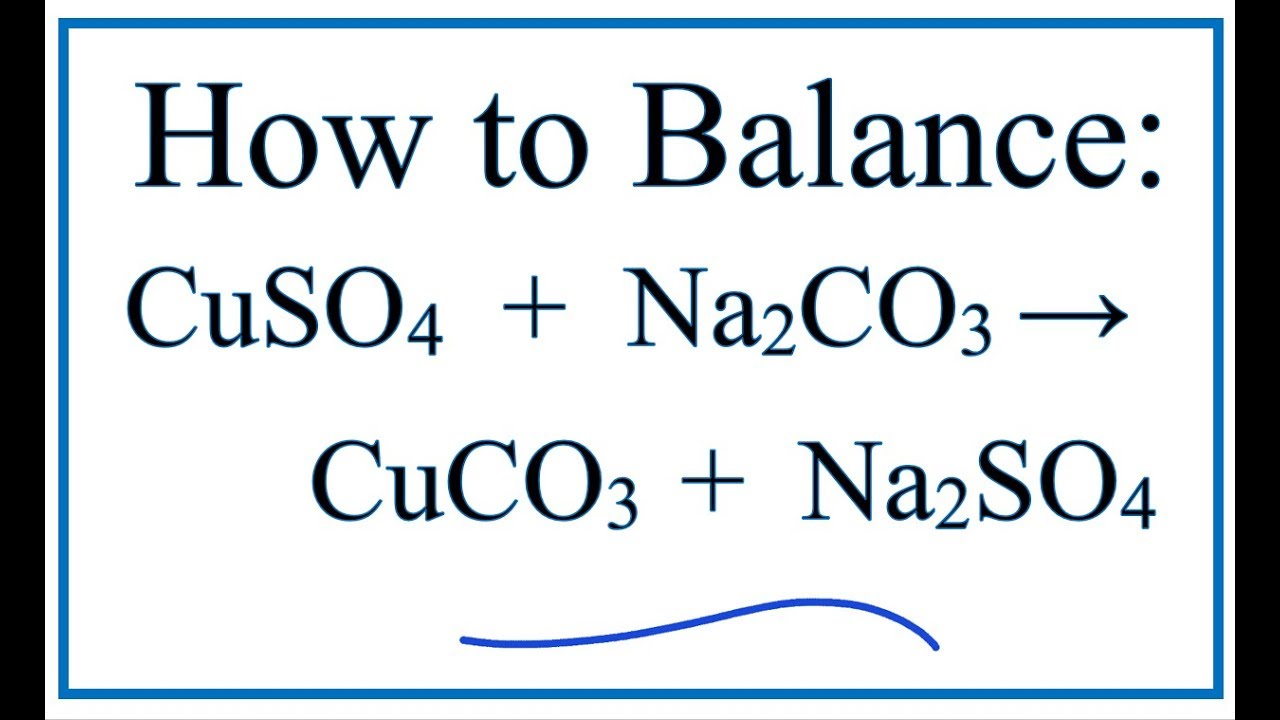
SOLVED: 1,. Convert the following word equations into balanced chemical equations aluminum + copper (II) sulfate a copper + aluminum sulfate b) sodium bicarbonate a sodium carbonate + water + carbon dioxide

What is the molecular, ionic, and net ionic equations for Copper(II) Sulfate + Sodium Phosphate? | Homework.Study.com
What happens when sodium hydroxide and copper sulfate are combined? Is the reaction dangerous? - Quora

Pipette adding sodium carbonate nickel sulfate. Nickel carbonate precipitate is formed result. Double displacement reaction Stock Photo - Alamy
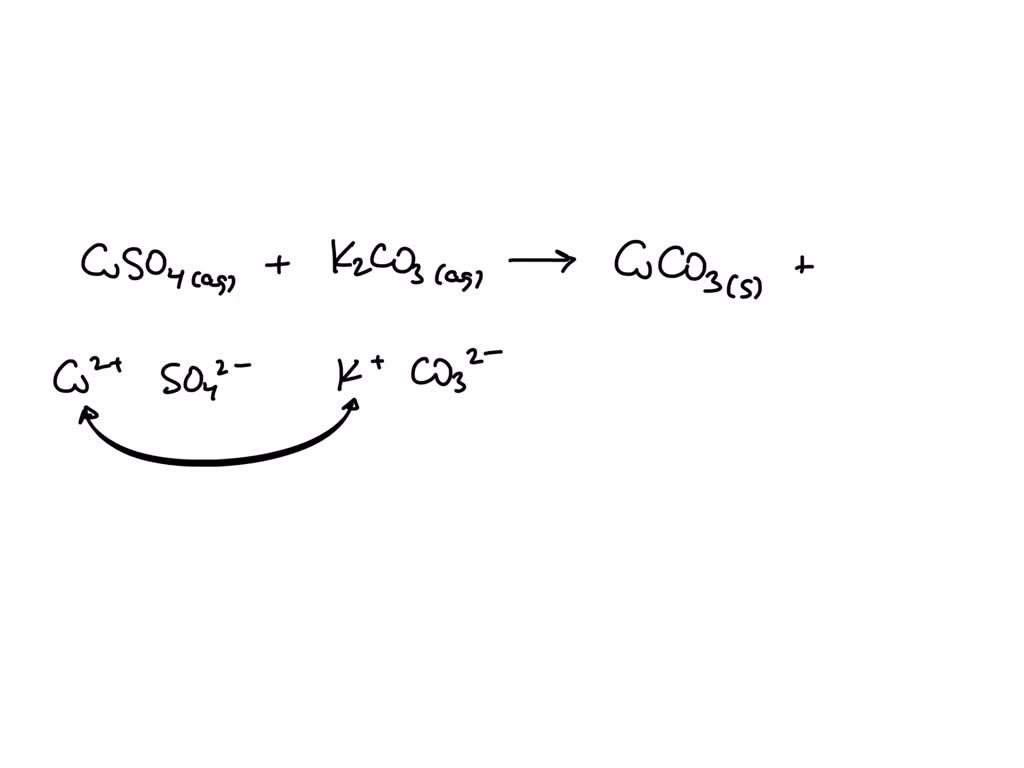
SOLVED: Aqueous solutions of copper (II) sulfate, CuSO4, and potassium carbonate, K2CO3, a blue parcipitate of copper (II) carbonate forms in addition to one other compound. We have to write the balanced
![Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2021/05/mq2-3.jpg?w=640)
Chemical Reactions to make copper(II) sulfate| Chemistry Basics [Online Video] – O Level Secondary Chemistry Tuition
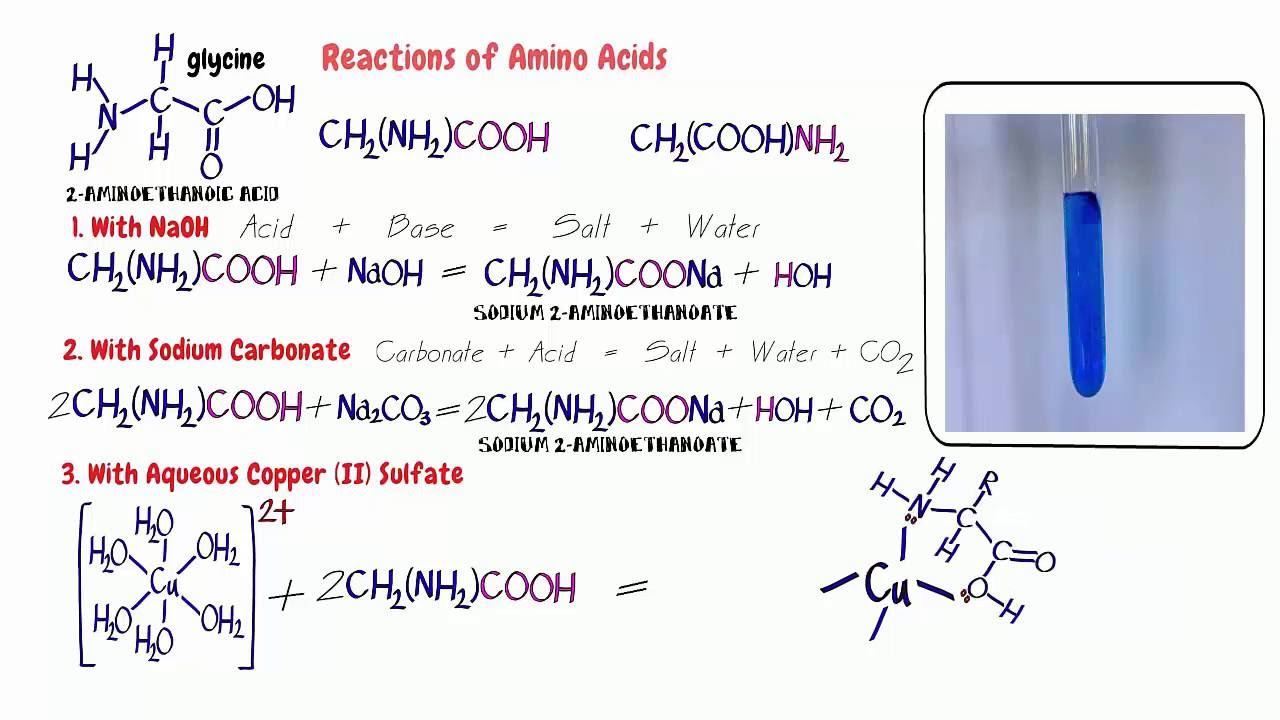
Amino Acids 3. Reactions with sodium hydroxide, sodium carbonate & copper (II) sulfate solution. - YouTube