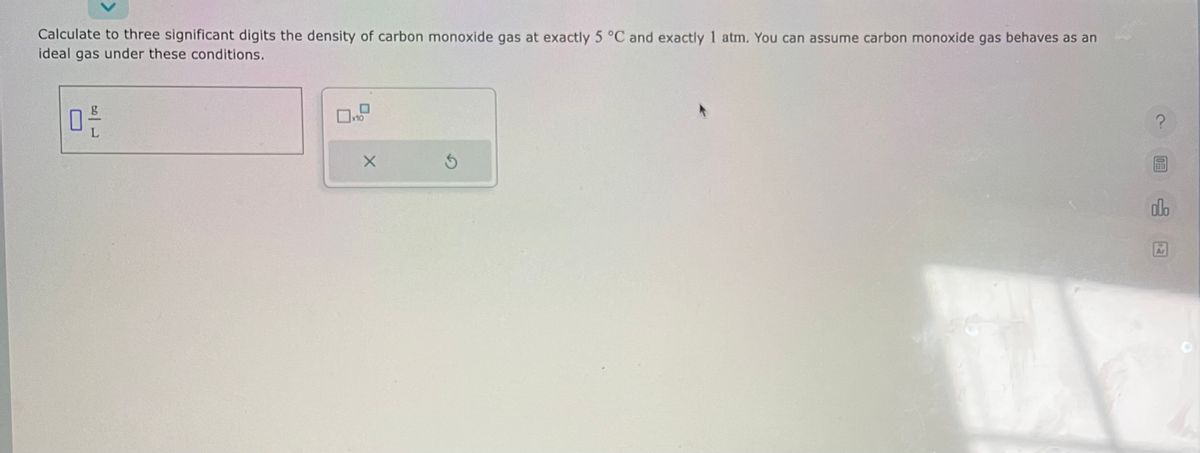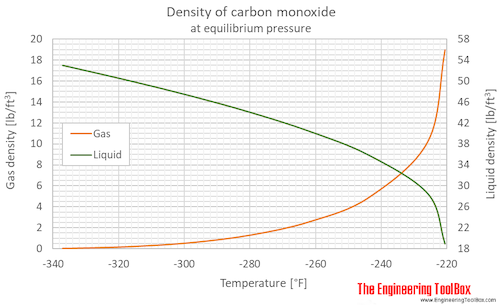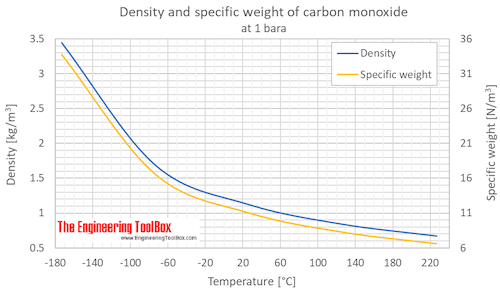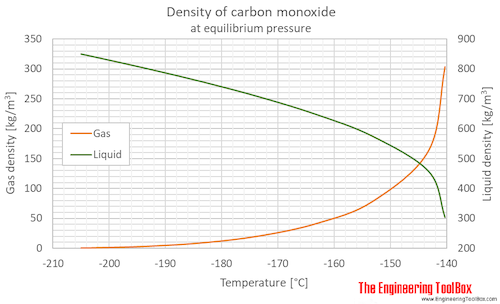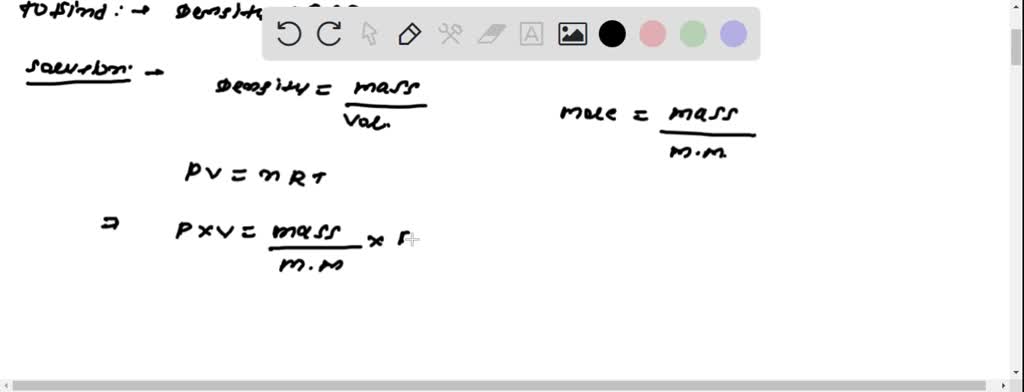
SOLVED: Calculate to three significant digits the density of carbon monoxide gas at exactly 20 degrees celsius and exactly 1 atm. You can assume carbon monoxide gas behaves as an ideal gas

SOLVED: What is the density of carbon monoxide gas at STP? STP: Standard Temperature and Pressure (T= 0 %C , Pz 1 atm) Select one: A. 1.25 g/L B 1.96 g/L C

OneClass: Use the molar volume of a gas at STP to calculate the density (in g/L ) of carbon monoxide ...

SOLVED: Calculate to three significant digits the density of dinitrogen monoxide gas at exactly 35°C and exactly 1atm. You can assume dinitrogen monoxide gas behaves as an ideal gas under these conditions.

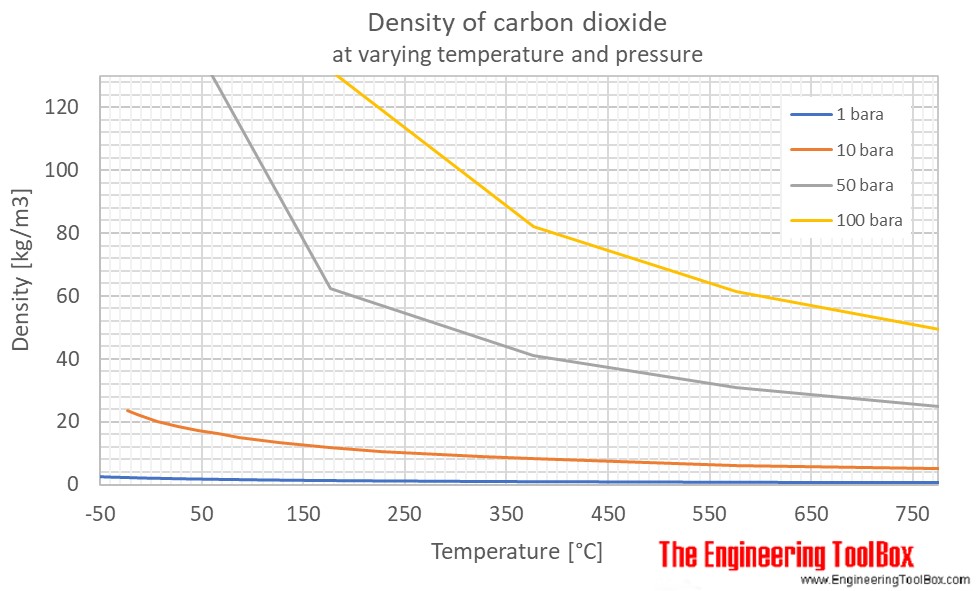
Density (kg/m3) of Carbon Dioxide as a Function of Temperature and Pressure - Carbon Dioxide Thermodynamic Properties Handbook - Wiley Online Library