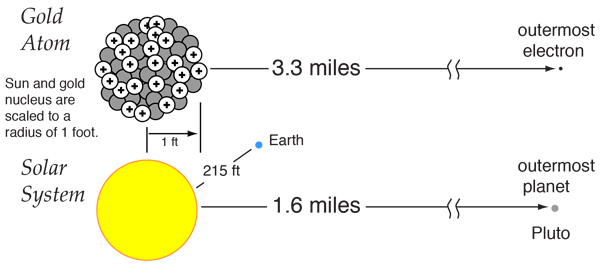
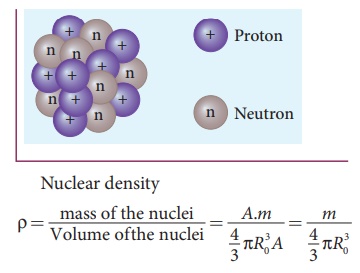
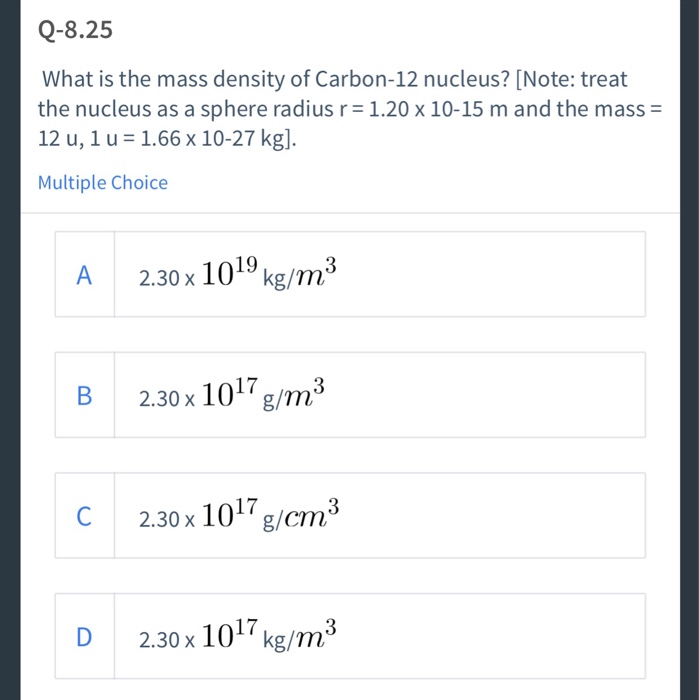
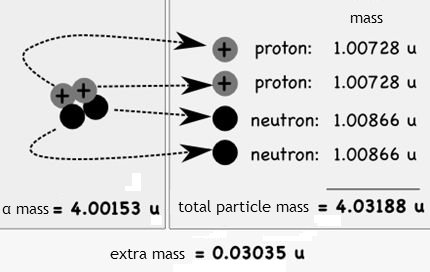
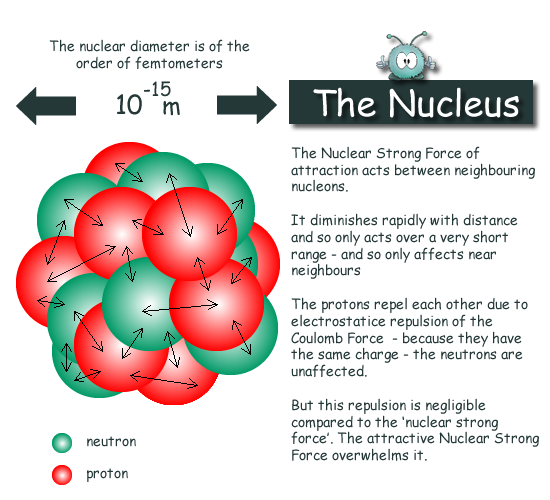
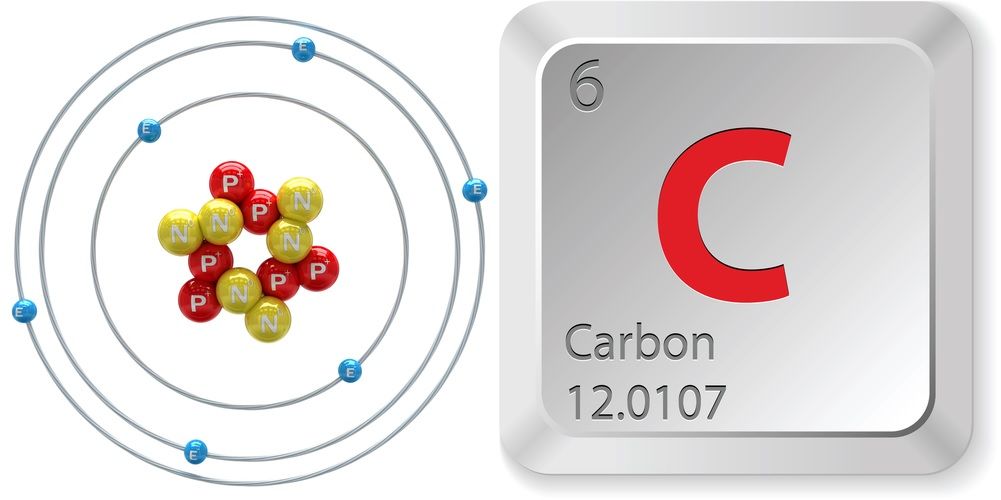
Find the density of `._(6)^(12)C` nucleus. Take atomic mass of `._(6)^(12)C` as `12.00 am u` Tak... - YouTube

Electron density at the BCP of C−H bonds against the electric field at... | Download Scientific Diagram
Electron density at the BCP of C−H bonds against the electric field at... | Download Scientific Diagram
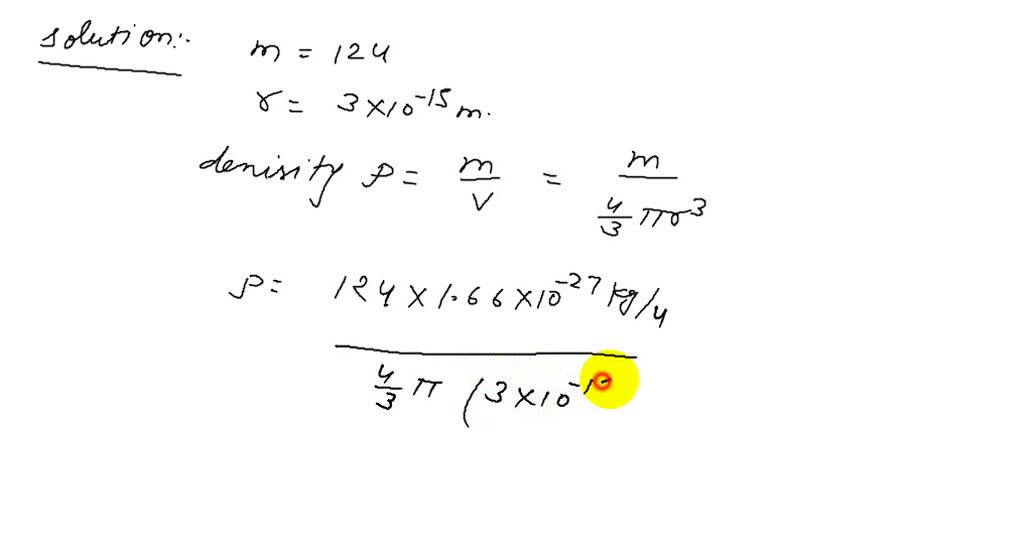
SOLVED:The radius of a carbon nucleus is about 3 ×10^-15 m and its mass is 12 u . Find the average density of the nuclear material. How many more times dense than

The radius of a carbon nucleus is about 3*10^-15m and its mass is 12amu. What is the average density of the nuclear material? And how many more times dense than water is

The density of a nucleus in which mass of each nucleon is 1.67 × 10^-27 kg and R0 = 1.4 × 10 ^-15 m is :
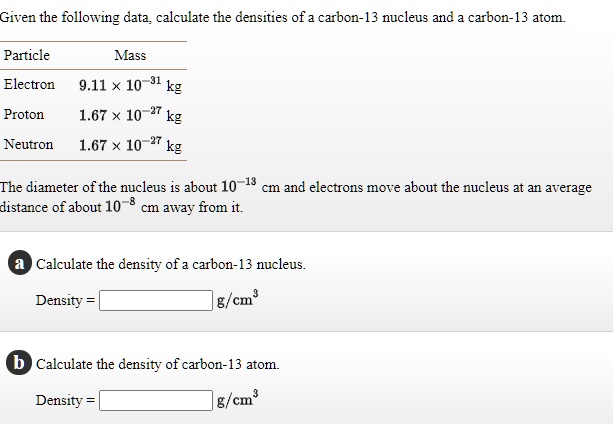
SOLVED: Given the following data calculate the densities of carbon- nucleus and carbon-13 atom Particle Mass Electron 9.11 * 10 kg 1.67 X 10 27 kg 1.67 x 10 kg Proton Neutron
![There's a lot of free volume! Density of carbon (as diamond) = 3 g/mL Density of 12 C nucleus = R nucleus = [(protons + neutrons) 1/3 ][1.2 cm] - ppt download There's a lot of free volume! Density of carbon (as diamond) = 3 g/mL Density of 12 C nucleus = R nucleus = [(protons + neutrons) 1/3 ][1.2 cm] - ppt download](https://slideplayer.com/6505017/22/images/slide_1.jpg)