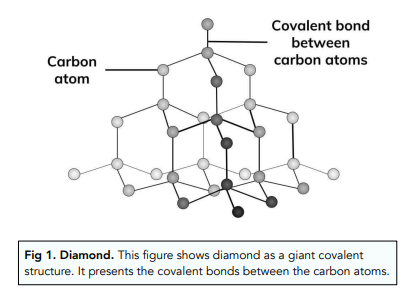
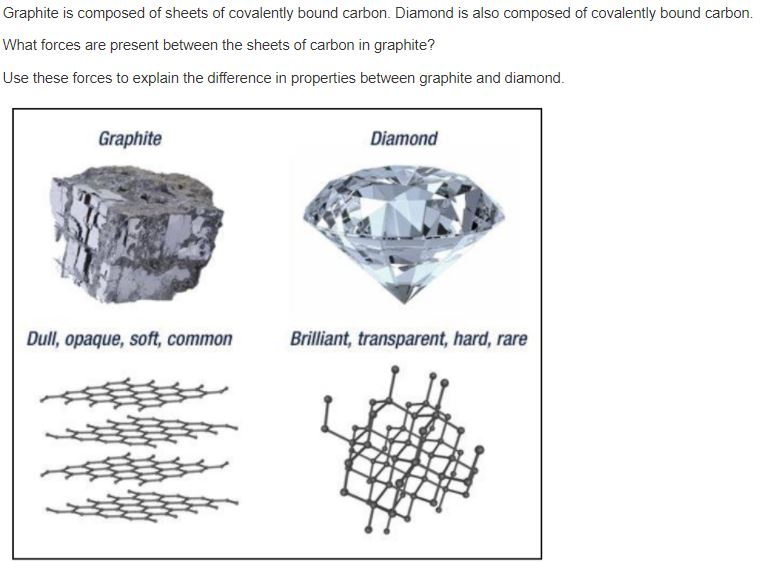
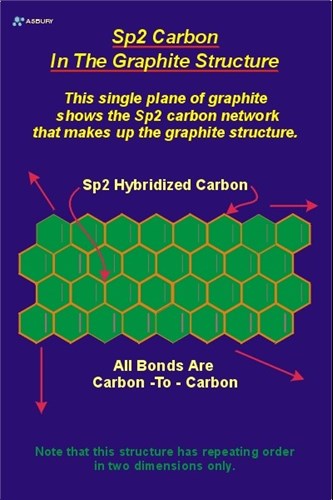
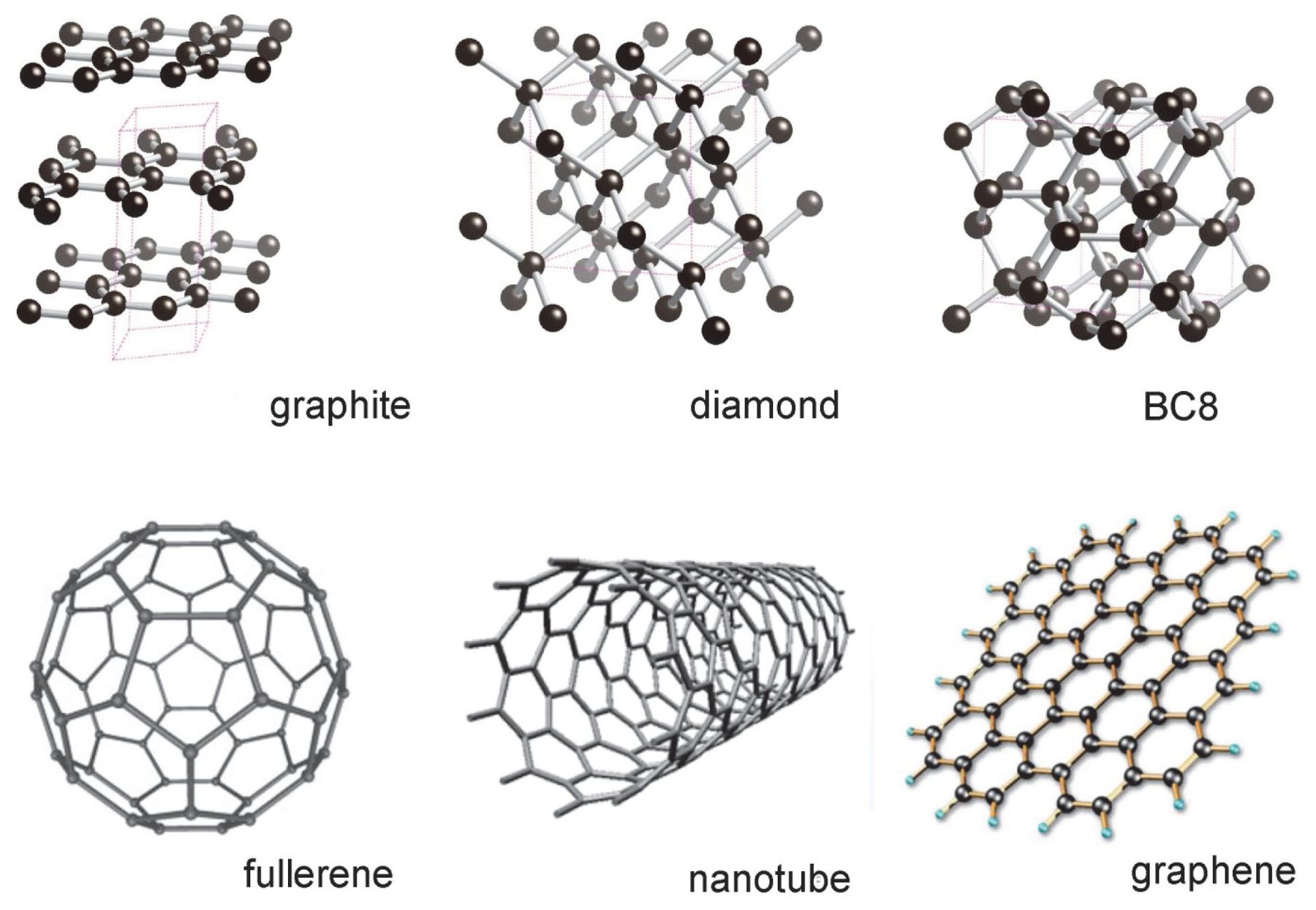
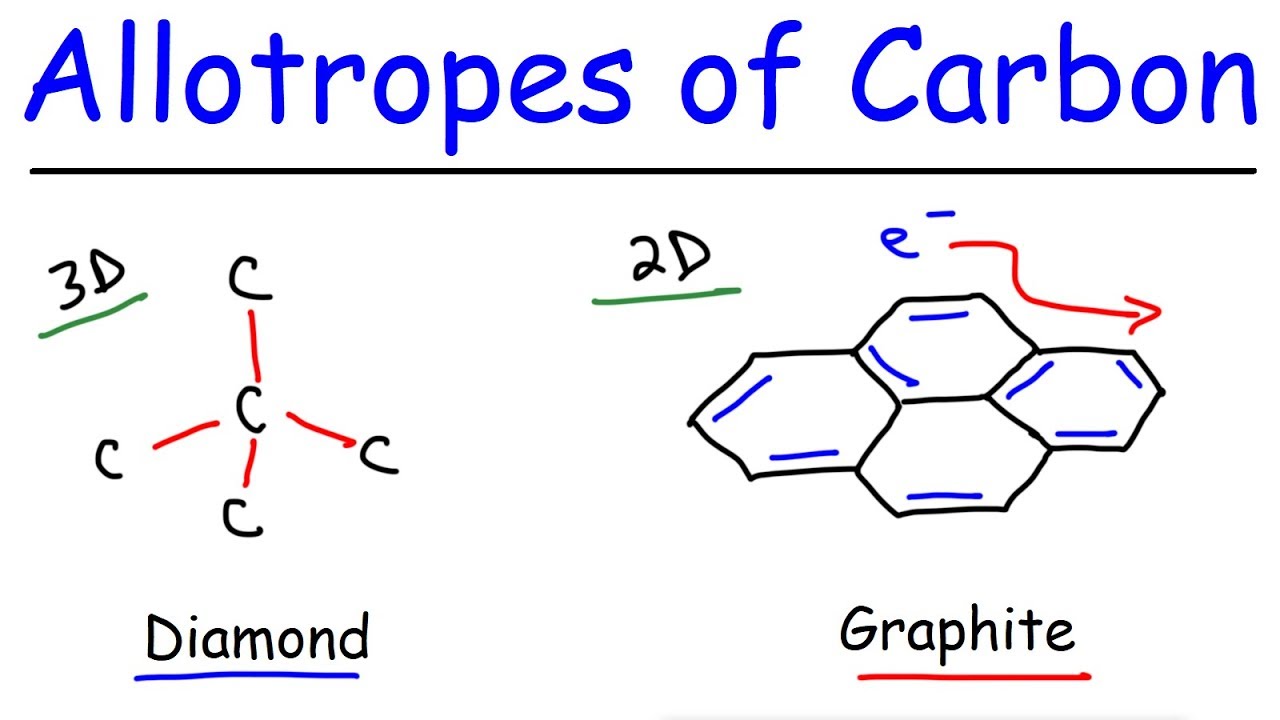
diamond and graphite both are allotropes of carbon.they are entirely different.make a table which shows the - Brainly.in

Explain the difference: A. Diamond and graphite. B. Crystalline and non-crystalline forms of carbon.

Explain the difference between diamond and graphite that what makes the graphite opaque to light while diamond is transparent to - Science - Carbon and its Compounds - 10691577 | Meritnation.com




.jpg)